-
 Primary Multi-Systemic Metastases in Osteosarcoma: Presentation, Treatment, and Survival of 83 Patients of the Cooperative Osteosarcoma Study Group
Primary Multi-Systemic Metastases in Osteosarcoma: Presentation, Treatment, and Survival of 83 Patients of the Cooperative Osteosarcoma Study Group -
 Addressing Genetic Tumor Heterogeneity, Post-Therapy Metastatic Spread, Cancer Repopulation, and Development of Acquired Tumor Cell Resistance
Addressing Genetic Tumor Heterogeneity, Post-Therapy Metastatic Spread, Cancer Repopulation, and Development of Acquired Tumor Cell Resistance -
 The Role of Macrophages in Sarcoma Tumor Microenvironment and Treatment
The Role of Macrophages in Sarcoma Tumor Microenvironment and Treatment -
 Synergistic Sensitization of High-Grade Serous Ovarian Cancer Cells Lacking Caspase-8 Expression to Chemotherapeutics Using Combinations of Small-Molecule BRD4 and CDK9 Inhibitors
Synergistic Sensitization of High-Grade Serous Ovarian Cancer Cells Lacking Caspase-8 Expression to Chemotherapeutics Using Combinations of Small-Molecule BRD4 and CDK9 Inhibitors
Journal Description
Cancers
Cancers
is a peer-reviewed, open access journal of oncology, published semimonthly online by MDPI. The Irish Association for Cancer Research (IACR), Spanish Association for Cancer Research (ASEICA), Biomedical Research Centre (CIBM), British Neuro-Oncology Society (BNOS) and Spanish Group for Cancer Immuno-Biotherapy (GÉTICA) are affiliated with Cancers and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q2 (Oncology) / CiteScore - Q1 (Oncology)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 17.9 days after submission; acceptance to publication is undertaken in 2.8 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Sections: published in 18 topical sections.
- Companion journals for Cancers include: Radiation and Onco.
Impact Factor:
5.2 (2022);
5-Year Impact Factor:
5.6 (2022)
Latest Articles
Bovine Meat and Milk Factor-like Sequences Are Frequently Detected in Renal Cell Carcinoma Tissues
Cancers 2024, 16(9), 1746; https://doi.org/10.3390/cancers16091746 (registering DOI) - 29 Apr 2024
Abstract
Previous studies have indicated a potential role of diet in the pathogenesis of renal cell carcinoma (RCC). Recently, circular bovine meat and milk factor (BMMF) DNAs have been identified in peritumoral tissues of human colon and breast cancers. Here, we investigated the prevalence
[...] Read more.
Previous studies have indicated a potential role of diet in the pathogenesis of renal cell carcinoma (RCC). Recently, circular bovine meat and milk factor (BMMF) DNAs have been identified in peritumoral tissues of human colon and breast cancers. Here, we investigated the prevalence of the DNA of these novel human pathogenic infectious agents in RCC and adjacent peritumoral renal tissues. DNA was extracted from formalin-fixed and paraffin-embedded (FFPE) RCC and peritumoral kidney tissues, including a test (n = 11) and a validation (n = 152) collection. BMMF1 and BMMF2 consensus primers were designed to screen for the presence of BMMF1- and BMMF2-like DNA. In addition, BMMF-specific PCR was performed on selected cases to test for the presence of additional regions of BMMF1 and BMMF2 genomes. A reference collection of hepatocellular carcinomas (HCCs; n = 60) and adjacent peritumoral liver tissues (n = 50) was also included. Our results demonstrated that BMMF1 and BMMF2 DNAs are frequently found in human RCC tissues and are particularly more prevalent in peritumoral kidney tissues. Of note, BMMF1 and BMMF2 genotype heterogeneity was higher in peritumoral kidney tissues compared to RCC tissues. This is the first study to directly test human FFPE tissues for BMMF1- and BMMF2-like DNA using consensus PCR and demonstrate BMMF DNA in neoplastic and peritumoral kidney tissues. The findings are in line with the recently proposed indirect etiopathogenetic role of BMMFs in, e.g., colorectal carcinogenesis. Follow-up studies are needed to explore the potential role of BMMFs in the etiopathogenesis of RCC.
Full article
(This article belongs to the Section Methods and Technologies Development)
Open AccessArticle
Incident Cancer Risk in Patients with Incident Type 2 Diabetes Mellitus in Hungary (Part 1)
by
Zsolt Abonyi-Tóth, György Rokszin, Ibolya Fábián, Zoltán Kiss, György Jermendy, Péter Kempler, Csaba Lengyel, István Wittmann, Gergő A. Molnár and Gábor Sütő
Cancers 2024, 16(9), 1745; https://doi.org/10.3390/cancers16091745 (registering DOI) - 29 Apr 2024
Abstract
(1) Background: Patients with type 2 diabetes mellitus (T2DM) are at higher risk of cancer but how these two diseases associate is still debated. The goal of this study was the assessment of the overall incidence of cancer among patients with newly diagnosed
[...] Read more.
(1) Background: Patients with type 2 diabetes mellitus (T2DM) are at higher risk of cancer but how these two diseases associate is still debated. The goal of this study was the assessment of the overall incidence of cancer among patients with newly diagnosed T2DM in Hungary. (2) Methods: A nationwide, retrospective, longitudinal study was performed using a Hungarian database. After exclusion of cases of age < 18 years, with gestational diabetes, with polycystic ovary syndrome, and with type 1 and prevalent type 2 diabetes mellitus, the incident T2DM (approx. 50,000 cases yearly) and for comparison, the diabetes-free Hungarian adult population (approx. 7,000,000 cases yearly) was included in the study. The primary endpoints were the overall and site-specific incidence and annual percentage change of the incidence of cancer in both populations. (3) Results: The overall incidence of cancer in patients amounted to 29.4/1000 and 6.6/1000 with or without T2DM, respectively, and the OR (95%CI) of cancer of the T2DM group was 4.32 (4.14–4.53), p < 0.0001. The risk of having cancer was age dependent. The incidence of cancer was declining in the non-diabetic but was unchanged in the T2DM population. The average lag time of diagnosing cancer after the detection of T2DM was 3.86 months. (4) Conclusions: Incident T2DM is associated with a significantly higher overall risk of incident cancer, with a reverse correlation of age. Newly registered T2DM patients were suggested to be screened for cancer within 6 months.
Full article
(This article belongs to the Special Issue Cancer and Diabetes: What Connections Lie between Them?)
Open AccessArticle
Evolution of Complexity of Palliative Care Needs and Patient Profiles According to the PALCOM Scale (Part Two): Pooled Analysis of the Cohorts for the Development and Validation of the PALCOM Scale in Advanced Cancer Patients
by
Albert Tuca, Margarita Viladot, Gemma Carrera, Lucia Llavata, Carmen Barrera, Manoli Chicote, Javier Marco-Hernández, Joan Padrosa, Carles Zamora-Martínez, Ignacio Grafia, Anais Pascual, Carme Font and Elena Font
Cancers 2024, 16(9), 1744; https://doi.org/10.3390/cancers16091744 (registering DOI) - 29 Apr 2024
Abstract
Introduction: Identifying the complexity of palliative care needs is a key aspect of referral to specialized multidisciplinary early palliative care (EPC) teams. The PALCOM scale is an instrument consisting of five multidimensional assessment domains developed in 2018 and validated in 2023 to identify
[...] Read more.
Introduction: Identifying the complexity of palliative care needs is a key aspect of referral to specialized multidisciplinary early palliative care (EPC) teams. The PALCOM scale is an instrument consisting of five multidimensional assessment domains developed in 2018 and validated in 2023 to identify the level of complexity in patients with advanced cancer. (1) Objectives: The main objective of this study was to determine the degree of instability (likelihood of level change or death), health resource consumption and the survival of patients according to the level of palliative complexity assigned at the baseline visit during a 6-month follow-up. (2) Method: An observational, prospective, multicenter study was conducted using pooled data from the development and validation cohort of the PALCOM scale. The main outcome variables were as follows: (a) instability ratio (IR), defined as the probability of level change or death; (b) emergency department visits; (c) days of hospitalization; (d) hospital death; (e) survival. All the variables were analyzed monthly according to the level of complexity assigned at the baseline visit. (3) Results: A total of 607 patients with advanced cancer were enrolled. According to the PALCOM scale, 20% of patients were classified as low complexity, 50% as medium and 30% as high complexity. The overall IR was 45% in the low complexity group, 68% in the medium complexity group and 78% in the high complexity group (p < 0.001). No significant differences in mean monthly emergency department visits (0.2 visits/ patient/month) were observed between the different levels of complexity. The mean number of days spent in hospital per month was 1.5 in the low complexity group, 1.8 in the medium complexity group and 3.2 in the high complexity group (p < 0.001). The likelihood of in-hospital death was significantly higher in the high complexity group (29%) compared to the medium (16%) and low (8%) complexity groups (p < 0.001). Six-month survival was significantly lower in the high complexity group (24%) compared to the medium (37%) and low (57%) complexity groups (p < 0.001). Conclusion: According to the PALCOM scale, more complex cases are associated with greater instability and use of hospital resources and lower survival. The data also confirm that the PALCOM scale is a consistent and useful tool for describing complexity profiles, targeting referrals to the EPC and managing the intensity of shared care.
Full article
(This article belongs to the Section Cancer Survivorship and Quality of Life)
Open AccessArticle
Quantitative Assessment of Tumor Contact with Neurogenic Zones and Its Effects on Survival: Insights beyond Traditional Predictors
by
Kirsten Jung, Johanna Kempter, Georg Prokop, Tim Herrmann, Michael Griessmair, Su-Hwan Kim, Claire Delbridge, Bernhard Meyer, Denise Bernhardt, Stephanie E. Combs, Claus Zimmer, Benedikt Wiestler, Friederike Schmidt-Graf and Marie-Christin Metz
Cancers 2024, 16(9), 1743; https://doi.org/10.3390/cancers16091743 (registering DOI) - 29 Apr 2024
Abstract
So far, the cellular origin of glioblastoma (GBM) needs to be determined, with prevalent theories suggesting emergence from transformed endogenous stem cells. Adult neurogenesis primarily occurs in two brain regions: the subventricular zone (SVZ) and the subgranular zone (SGZ) of the hippocampal dentate
[...] Read more.
So far, the cellular origin of glioblastoma (GBM) needs to be determined, with prevalent theories suggesting emergence from transformed endogenous stem cells. Adult neurogenesis primarily occurs in two brain regions: the subventricular zone (SVZ) and the subgranular zone (SGZ) of the hippocampal dentate gyrus. Whether the proximity of GBM to these neurogenic niches affects patient outcome remains uncertain. Previous studies often rely on subjective assessments, limiting the reliability of those results. In this study, we assessed the impact of GBM’s relationship with the cortex, SVZ and SGZ on clinical variables using fully automated segmentation methods. In 177 glioblastoma patients, we calculated optimal cutpoints of minimal distances to the SVZ and SGZ to distinguish poor from favorable survival. The impact of tumor contact with neurogenic zones on clinical parameters, such as overall survival, multifocality, MGMT promotor methylation, Ki-67 and KPS score was also examined by multivariable regression analysis, chi-square test and Mann–Whitney-U. The analysis confirmed shorter survival in tumors contacting the SVZ with an optimal cutpoint of 14 mm distance to the SVZ, separating poor from more favorable survival. In contrast, tumor contact with the SGZ did not negatively affect survival. We did not find significant correlations with multifocality or MGMT promotor methylation in tumors contacting the SVZ, as previous studies discussed. These findings suggest that the spatial relationship between GBM and neurogenic niches needs to be assessed differently. Objective measurements disprove prior assumptions, warranting further research on this topic.
Full article
(This article belongs to the Section Cancer Survivorship and Quality of Life)
Open AccessArticle
Long-Term Results of Stereotactic Radiotherapy in Patients with at Least 10 Brain Metastases at Diagnosis
by
Rémy Kinj, Andreas Felix Hottinger, Till Tobias Böhlen, Mahmut Ozsahin, Véronique Vallet, Vincent Dunet, Hasna Bouchaab, Solange Peters, Constantin Tuleasca, Jean Bourhis and Luis Schiappacasse
Cancers 2024, 16(9), 1742; https://doi.org/10.3390/cancers16091742 (registering DOI) - 29 Apr 2024
Abstract
Purpose: to evaluate an SRT approach in patients with at least 10 lesions at the time of BM initial diagnosis. Methods: This is a monocentric prospective cohort of patients treated by SRT, followed by a brain MRI every two months. Subsequent SRT could
[...] Read more.
Purpose: to evaluate an SRT approach in patients with at least 10 lesions at the time of BM initial diagnosis. Methods: This is a monocentric prospective cohort of patients treated by SRT, followed by a brain MRI every two months. Subsequent SRT could be delivered in cases of new BMs during follow-up. The main endpoints were local control rate (LCR), overall survival (OS), and strategy success rate (SSR). Acute and late toxicity were evaluated. Results: Seventy patients were included from October 2014 to January 2019, and the most frequent primary diagnosis was non-small-cell lung cancer (N = 36, 51.4%). A total of 1174 BMs were treated at first treatment, corresponding to a median number of 14 BMs per patient. Most of the patients (N = 51, 72.6%) received a single fraction of 20–24 Gy. At 1 year, OS was 62.3%, with a median OS of 19.2 months, and SSR was 77.8%. A cumulative number of 1537 BM were treated over time, corresponding to a median cumulative number of 16 BM per patient. At 1-year, the LCR was 97.3%, with a cumulative incidence of radio-necrosis of 2.1% per lesion. Three patients (4.3%) presented Grade 2 toxicity, and there was no Grade ≥ 3 toxicity. The number of treated BMs and the treatment volume did not influence OS or SSR (p > 0.05). Conclusions: SRT was highly efficient in controlling the BM, with minimal side effects. In this setting, an SRT treatment should be proposed even in patients with ≥10 BMs at diagnosis.
Full article
(This article belongs to the Special Issue Stereotactic Radiotherapy in Tumor Ablation (Volume II))
Open AccessReview
Gastric Cancer Surgery: Balancing Oncological Efficacy against Postoperative Morbidity and Function Detriment
by
Andrianos Tsekrekos, Yasuhiro Okumura, Ioannis Rouvelas and Magnus Nilsson
Cancers 2024, 16(9), 1741; https://doi.org/10.3390/cancers16091741 (registering DOI) - 29 Apr 2024
Abstract
Significant progress has been made in the surgical management of gastric cancer over the years, and previous discrepancies in surgical practice between different parts of the world have gradually lessened. A transition from the earlier period of progressively more extensive surgery to the
[...] Read more.
Significant progress has been made in the surgical management of gastric cancer over the years, and previous discrepancies in surgical practice between different parts of the world have gradually lessened. A transition from the earlier period of progressively more extensive surgery to the current trend of a more tailored and evidence-based approach is clear. Prophylactic resection of adjacent anatomical structures or neighboring organs and extensive lymph node dissections that were once assumed to increase the chances of long-term survival are now performed selectively. Laparoscopic gastrectomy has been widely adopted and its indications have steadily expanded, from early cancers located in the distal part of the stomach, to locally advanced tumors where total gastrectomy is required. In parallel, function-preserving surgery has also evolved and now constitutes a valid option for early gastric cancer. Pylorus-preserving and proximal gastrectomy have improved the postoperative quality of life of patients, and sentinel node navigation surgery is being explored as the next step in the process of further refining the minimally invasive concept. Moreover, innovative techniques such as indocyanine green fluorescence imaging and robot-assisted gastrectomy are being introduced in clinical practice. These technologies hold promise for enhancing surgical precision, ultimately improving the oncological and functional outcomes.
Full article
(This article belongs to the Special Issue Gastric Cancer: Evolving Landscape and Emerging Therapies)
Open AccessArticle
The Molecular Landscape of Primary CNS Lymphomas (PCNSLs) in Children and Young Adults
by
Zhi-Feng Shi, Kay Ka-Wai Li, Anthony Pak-Yin Liu, Nellie Yuk-Fei Chung, Sze-Ching Wong, Hong Chen, Peter Yat-Ming Woo, Danny Tat-Ming Chan, Ying Mao and Ho-Keung Ng
Cancers 2024, 16(9), 1740; https://doi.org/10.3390/cancers16091740 - 29 Apr 2024
Abstract
Pediatric brain tumors are often noted to be different from their adult counterparts in terms of molecular features. Primary CNS lymphomas (PCNSLs) are mostly found in elderly adults and are uncommon in children and teenagers. There has only been scanty information about the
[...] Read more.
Pediatric brain tumors are often noted to be different from their adult counterparts in terms of molecular features. Primary CNS lymphomas (PCNSLs) are mostly found in elderly adults and are uncommon in children and teenagers. There has only been scanty information about the molecular features of PCNSLs at a young age. We examined PCNSLs in 34 young patients aged between 7 and 39 years for gene rearrangements of BCl2, BCL6, CCND1, IRF4, IGH, IGL, IGK, and MYC, homozygous deletions (HD) of CDKN2A, and HLA by FISH. Sequencing was performed using WES, panel target sequencing, or Sanger sequencing due to the small amount of available tissues. The median OS was 97.5 years and longer than that for older patients with PCNSLs. Overall, only 14 instances of gene rearrangement were found (5%), and patients with any gene rearrangement were significantly older (p = 0.029). CDKN2A HD was associated with a shorter OS (p < 0.001). Only 10/31 (32%) showed MYD88 mutations, which were not prognostically significant, and only three of them were L265P mutations. CARD11 mutations were found in 8/24 (33%) cases only. Immunophenotypically, the cases were predominantly GCB, in contrast to older adults (61%). In summary, we showed that molecular findings identified in the PCNSLs of the older patients were only sparingly present in pediatric and young adult patients.
Full article
(This article belongs to the Special Issue Molecular Pathology of Brain Tumors)
Open AccessReview
Reappraisal of the Roles of the Sonic Hedgehog Signaling Pathway in Hepatocellular Carcinoma
by
Kuo-Shyang Jeng, Chiung-Fang Chang, Yuk-Ming Tsang, I-Shyan Sheen and Chi-Juei Jeng
Cancers 2024, 16(9), 1739; https://doi.org/10.3390/cancers16091739 - 29 Apr 2024
Abstract
HCC remains one of the leading causes of cancer-related death globally. The main challenges in treatments of hepatocellular carcinoma (HCC) primarily arise from high rates of postoperative recurrence and the limited efficacy in treating advanced-stage patients. Various signaling pathways involved in HCC have
[...] Read more.
HCC remains one of the leading causes of cancer-related death globally. The main challenges in treatments of hepatocellular carcinoma (HCC) primarily arise from high rates of postoperative recurrence and the limited efficacy in treating advanced-stage patients. Various signaling pathways involved in HCC have been reported. Among them, the Sonic hedgehog (SHH) signaling pathway is crucial. The presence of SHH ligands is identified in approximately 60% of HCC tumor tissues, including tumor nests. PTCH-1 and GLI-1 are detected in more than half of HCC tissues, while GLI-2 is found in over 84% of HCC tissues. The SHH signaling pathway (including canonical and non-canonical) is involved in different aspects of HCC, including hepatocarcinogenesis, tumor growth, tumor invasiveness, progression, and migration. The SHH signaling pathway also contributes to recurrence, metastasis, modulation of the cancer microenvironment, and sustaining cancer stem cells. It also affects the resistance of HCC cells to chemotherapy, target therapy, and radiotherapy. Reappraisal of the roles of the SHH signaling pathway in HCC may trigger some novel therapies for HCC.
Full article
(This article belongs to the Section Molecular Cancer Biology)
Open AccessArticle
The Impact of the COVID-19 Pandemic on Time to Treatment in Surgical Oncology: A National Registry Study in the Netherlands
by
Roos M. G. van Vuren, Yester F. Janssen, Rianne N. M. Hogenbirk, Michelle R. de Graaff, Rinske van den Hoek, Schelto Kruijff, David J. Heineman, Willemijn Y. van der Plas and Michel W. J. M. Wouters
Cancers 2024, 16(9), 1738; https://doi.org/10.3390/cancers16091738 - 29 Apr 2024
Abstract
To avoid delay in oncological treatment, a 6-weeks norm for time to treatment has been agreed on in the Netherlands. However, the impact of the COVID-19 pandemic on health systems resulted in reduced capacity for regular surgical care. In this study, we investigated
[...] Read more.
To avoid delay in oncological treatment, a 6-weeks norm for time to treatment has been agreed on in the Netherlands. However, the impact of the COVID-19 pandemic on health systems resulted in reduced capacity for regular surgical care. In this study, we investigated the impact of the COVID-19 pandemic on time to treatment in surgical oncology in the Netherlands. Methods: A population-based analysis of data derived from five surgical audits, including patients who underwent surgery for lung cancer, colorectal cancer, upper gastro-intestinal, and hepato-pancreato-biliary (HPB) malignancies, was performed. The COVID-19 cohort of 2020 was compared to the historic cohorts of 2018 and 2019. Primary endpoints were time to treatment initiation and the proportion of patients whose treatment started within 6 weeks. The secondary objective was to evaluate the differences in characteristics and tumour stage distribution between patients treated before and during the COVID-19 pandemic. Results: A total of 14,567 surgical cancer patients were included in this study, of these 3292 treatments were started during the COVID-19 pandemic. The median time to treatment decreased during the pandemic (26 vs. 27 days, p < 0.001) and the proportion of patients whose treatment started within 6 weeks increased (76% vs. 73%, p < 0.001). In a multivariate logistic regression analysis, adjusting for patient characteristics, no significant difference in post-operative outcomes between patients who started treatment before or after 6 weeks was found. Overall, the number of procedures performed per week decreased by 8.1% during the pandemic. This reduction was most profound for patients with stage I lung carcinoma and colorectal carcinoma. There were fewer patients with pulmonary comorbidities in the pandemic cohort (11% vs. 13%, p = 0.003). Conclusions: Despite pressure on the capacity of the healthcare system during the COVID-19 pandemic, a larger proportion of surgical oncological patients started treatment within six weeks, possibly due to prioritisation of cancer care and reductions in elective procedures. However, during the pandemic, a decrease in the number of surgical oncological procedures performed in the Netherlands was observed, especially for patients with stage I disease.
Full article
(This article belongs to the Collection The Impact of COVID-19 Infection in Cancer)
Open AccessArticle
High Serum Levels of CCL20 Are Associated with Recurrence and Unfavorable Overall Survival in Advanced Melanoma Patients Receiving Immunotherapy
by
Julian Kött, Inka Lilott Hoehne, Isabel Heidrich, Noah Zimmermann, Kim-Lea Reese, Tim Zell, Glenn Geidel, Alessandra Rünger, Stefan W. Schneider, Klaus Pantel, Daniel J. Smit and Christoffer Gebhardt
Cancers 2024, 16(9), 1737; https://doi.org/10.3390/cancers16091737 - 29 Apr 2024
Abstract
Background: Immune checkpoint inhibition has revolutionized melanoma therapy, but many patients show primary or secondary resistance. Biomarkers are, therefore, urgently required to predict response prior to the initiation of therapy and to monitor disease progression. Methods: In this prospective study, we analyzed the
[...] Read more.
Background: Immune checkpoint inhibition has revolutionized melanoma therapy, but many patients show primary or secondary resistance. Biomarkers are, therefore, urgently required to predict response prior to the initiation of therapy and to monitor disease progression. Methods: In this prospective study, we analyzed the serum C-C motif chemokine ligand 20 (CCL20) concentration using an enzyme-linked immunosorbent assay. Blood was obtained at baseline before the initiation of immunotherapy with anti-PD-1 monotherapy or Nivolumab and Ipilimumab in advanced melanoma patients (stages III and IV) enrolled at the University Medical Center Hamburg-Eppendorf. The CCL20 levels were correlated with clinico-pathological parameters and disease-related outcomes. Results: An increased C-C motif chemokine ligand 20 (CCL20) concentration (≥0.34 pg/mL) at baseline was associated with a significantly impaired progression-free survival (PFS) in the high-CCL20 group (3 months (95% CI: 2–6 months) vs. 11 months (95% CI: 6–26 months)) (p = 0.0033) and could be identified as an independent negative prognostic factor for PFS in univariate (Hazard Ratio (HR): 1.98, 95% CI 1.25–3.12, p = 0.004) and multivariate (HR: 1.99, 95% CI 1.21–3.29, p = 0.007) Cox regression analysis, which was associated with a higher risk than S100 (HR: 1.74). Moreover, high CCL20 levels were associated with impaired overall survival (median OS not reached for low-CCL20 group, p = 0.042) with an HR of 1.85 (95% CI 1.02–3.37, p = 0.043) in univariate analysis similar to the established prognostic marker S100 (HR: 1.99, 95% CI: 1.02–3.88, p = 0.043). Conclusions: CCL20 may represent a novel blood-based biomarker for the prediction of resistance to immunotherapy that can be used in combination with established strong clinical predictors (e.g., ECOG performance score) and laboratory markers (e.g., S100) in advanced melanoma patients. Future prospective randomized trials are needed to establish CCL20 as a liquid biopsy-based biomarker in advanced melanoma.
Full article
(This article belongs to the Special Issue Novel Developments on Skin Cancer Diagnostics and Treatment)
►▼
Show Figures
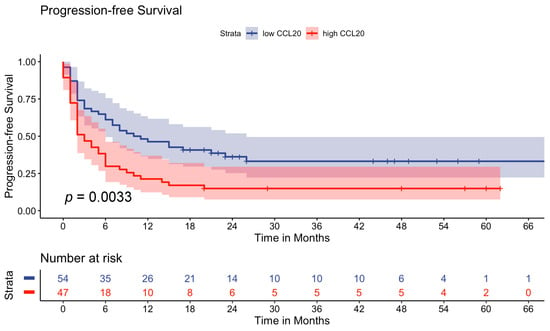
Figure 1
Open AccessArticle
Improving Breast Cancer Outcomes for Indigenous Women in Australia
by
Vita Christie, Lynette Riley, Deb Green, Janaki Amin, John Skinner, Chris Pyke and Kylie Gwynne
Cancers 2024, 16(9), 1736; https://doi.org/10.3390/cancers16091736 - 29 Apr 2024
Abstract
In Australia, the incidence rate of breast cancer is lower in Indigenous* women than non-Indigenous women; however, the mortality rate is higher, with Indigenous women 1.2 times more likely to die from the disease. This paper provides practical and achievable solutions to improve
[...] Read more.
In Australia, the incidence rate of breast cancer is lower in Indigenous* women than non-Indigenous women; however, the mortality rate is higher, with Indigenous women 1.2 times more likely to die from the disease. This paper provides practical and achievable solutions to improve health outcomes for Indigenous women with breast cancer in Australia. This research employed the Context–Mechanism–Outcome (CMO) framework to reveal potential mechanisms and contextual factors that influence breast cancer outcomes for Indigenous women, stratified into multiple levels, namely, micro (interpersonal), meso (systemic) and macro (policy) levels. The CMO framework allowed us to interpret evidence regarding Indigenous women and breast cancer and provides nine practical ways to improve health outcomes and survival rates.
Full article
(This article belongs to the Special Issue Quality and Clinical Outcomes Improvement in the Management of Oncology Patients)
►▼
Show Figures
Figure 1
Open AccessArticle
Management of High-Risk Neuroblastoma with Soft-Tissue-Only Disease in the Era of Anti-GD2 Immunotherapy
by
Maite Gorostegui, Juan Pablo Muñoz, Sara Perez-Jaume, Margarida Simao-Rafael, Cristina Larrosa, Moira Garraus, Noelia Salvador, Cinzia Lavarino, Lucas Krauel, Salvador Mañe, Alicia Castañeda and Jaume Mora
Cancers 2024, 16(9), 1735; https://doi.org/10.3390/cancers16091735 - 29 Apr 2024
Abstract
Neuroblastoma presents with two patterns of disease: locoregional or systemic. The poor prognostic risk factors of locoregional neuroblastoma (LR-NB) include age, MYCN or MDM2-CDK4 amplification, 11q, histology, diploidy with ALK or TERT mutations, and ATRX aberrations. Anti-GD2 immunotherapy has significantly improved the outcome
[...] Read more.
Neuroblastoma presents with two patterns of disease: locoregional or systemic. The poor prognostic risk factors of locoregional neuroblastoma (LR-NB) include age, MYCN or MDM2-CDK4 amplification, 11q, histology, diploidy with ALK or TERT mutations, and ATRX aberrations. Anti-GD2 immunotherapy has significantly improved the outcome of high-risk (HR) NB and is mostly effective against osteomedullary minimal residual disease (MRD), but less so against soft tissue disease. The question is whether adding anti-GD2 monoclonal antibodies (mAbs) benefits patients with HR-NB compounded by only soft tissue. We reviewed 31 patients treated at SJD for HR-NB with no osteomedullary involvement at diagnosis. All tumors had molecular genetic features of HR-NB. The outcome after first-line treatment showed 25 (80.6%) patients achieving CR. Thirteen patients remain in continued CR, median follow-up 3.9 years. We analyzed whether adding anti-GD2 immunotherapy to first-line treatment had any prognostic significance. The EFS analysis using Cox models showed a HR of 0.20, p = 0.0054, and an 80% decrease in the risk of relapse in patients treated with anti-GD2 immunotherapy in the first line. Neither EFS nor OS were significantly different by CR status after first-line treatment. In conclusion, adding treatment with anti-GD2 mAbs at the stage of MRD helps prevent relapse that unequivocally portends poor survival.
Full article
(This article belongs to the Section Pediatric Oncology)
►▼
Show Figures
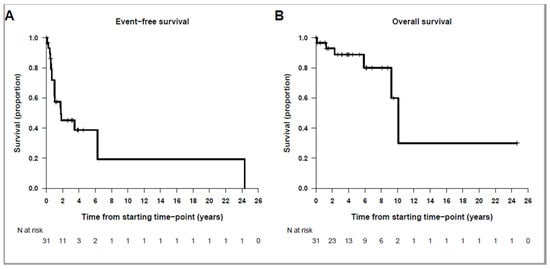
Figure 1
Open AccessReview
Treatment Strategies for Non-Small-Cell Lung Cancer with Comorbid Respiratory Disease; Interstitial Pneumonia, Chronic Obstructive Pulmonary Disease, and Tuberculosis
by
Ryota Otoshi, Satoshi Ikeda, Taichi Kaneko, Shinobu Sagawa, Chieri Yamada, Kosumi Kumagai, Asami Moriuchi, Akimasa Sekine, Tomohisa Baba and Takashi Ogura
Cancers 2024, 16(9), 1734; https://doi.org/10.3390/cancers16091734 - 29 Apr 2024
Abstract
Non-small cell lung cancer (NSCLC) patients are often complicated by other respiratory diseases, including interstitial pneumonia (IP), chronic obstructive pulmonary disease (COPD), and pulmonary tuberculosis (TB), and the management of which can be problematic. NSCLC patients with IP sometimes develop fatal acute exacerbation
[...] Read more.
Non-small cell lung cancer (NSCLC) patients are often complicated by other respiratory diseases, including interstitial pneumonia (IP), chronic obstructive pulmonary disease (COPD), and pulmonary tuberculosis (TB), and the management of which can be problematic. NSCLC patients with IP sometimes develop fatal acute exacerbation induced by pharmacotherapy, and the establishment of a safe treatment strategy is desirable. For advanced NSCLC with IP, carboplatin plus nanoparticle albumin-bound paclitaxel is a relatively safe and effective first-line treatment option. Although the safety of immune checkpoint inhibitors (ICIs) for these populations remains controversial, ICIs have the potential to provide long-term survival. The severity of COPD is an important prognostic factor in NSCLC patients. Although COPD complications do not necessarily limit treatment options, it is important to select drugs with fewer side effects on the heart and blood vessels as well as the lungs. Active TB is complicated by 2–5% of NSCLC cases during their disease course. Since pharmacotherapy, especially ICIs, reportedly induces the development of TB, the possibility of developing TB should always be kept in mind during NSCLC treatment. To date, there is no coherent review article on NSCLC with these pulmonary complications. This review article summarizes the current evidence and discusses future prospects for treatment strategies for NSCLC patients complicated with IP, severe COPD, and TB.
Full article
(This article belongs to the Section Cancer Therapy)
►▼
Show Figures
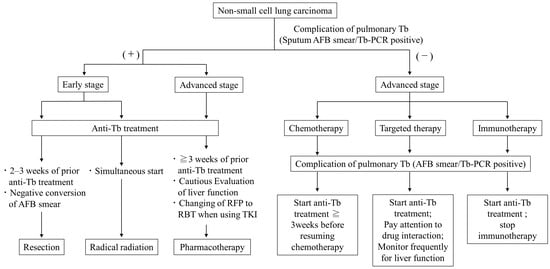
Figure 1
Open AccessArticle
A Role for iNOS in Erastin Mediated Reduction of P-Glycoprotein Transport Activity
by
Shalyn M. Brown, Birandra K. Sinha and Ronald E. Cannon
Cancers 2024, 16(9), 1733; https://doi.org/10.3390/cancers16091733 - 29 Apr 2024
Abstract
The blood–brain barrier is composed of both a physical barrier and an enzymatic barrier. Tight junction (TJ) proteins expressed between endothelial cells of brain capillaries provide the physical barrier to paracellular movement of ions and molecules to the brain, while luminal-facing efflux transporters
[...] Read more.
The blood–brain barrier is composed of both a physical barrier and an enzymatic barrier. Tight junction (TJ) proteins expressed between endothelial cells of brain capillaries provide the physical barrier to paracellular movement of ions and molecules to the brain, while luminal-facing efflux transporters enzymatically restrict the entry of blood-borne molecules from entering the brain. The expression and activity of ATP Binding Cassette transporters or “ABC” transporters in endothelial cells of the BBB and in human tumor cells are dynamically regulated by numerous signaling pathways. P-glycoprotein (P-gp), (ABCB1), is arguably the most studied transporter of the BBB, and in human cell lines. P-glycoprotein transport activity is rapidly inhibited by signaling pathways that call for the rapid production of nitric oxide (NO) from the inducible nitric oxide synthase enzyme, iNOS. This study investigated how nano-molar levels of the selective chemotherapeutic erastin affect the activity or expression of P-glycoprotein transporter in brain capillaries and in human tumor cell lines. We chose erastin because it signals to iNOS for NO production at low concentrations. Furthermore, erastin inhibits the cellular uptake of cystine through the XC− cystine/glutamate antiporter. Since previous reports indicate that NO production from iNOS can rapidly inhibit P-gp activity in tumor cells, we wondered if induction of iNOS by erastin could also rapidly reduce P-glycoprotein transport activity in brain endothelial cells and in human tumor cell lines. We show here that low concentrations of erastin (1 nM) can induce iNOS, inhibit the activity of P-glycoprotein, and reduce the intracellular uptake of cystine via the Xc- cystine/glutamate antiporter. Consistent with reduced P-glycoprotein activity in rat brain capillary endothelial cells, we show that human tumor cell lines exposed to erastin become more sensitive to cytotoxic substrates of P-glycoprotein.
Full article
(This article belongs to the Collection Innovations in Cancer Drug Development Research)
►▼
Show Figures
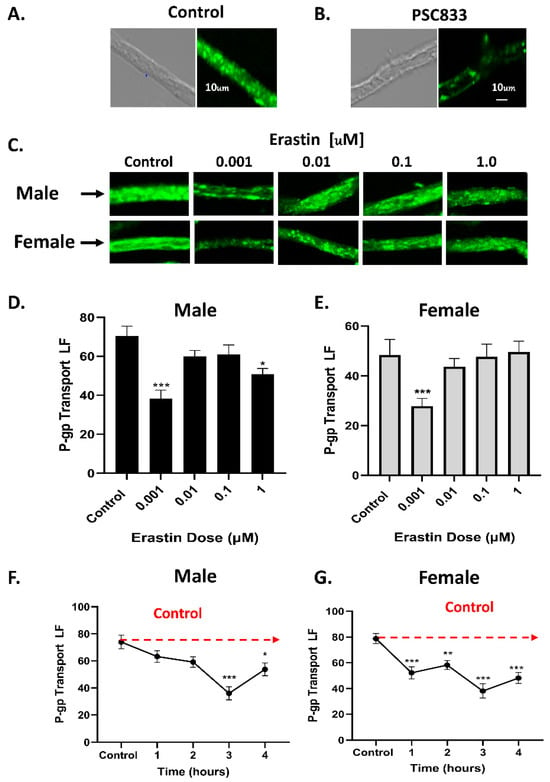
Figure 1
Open AccessReview
Efficacy and Safety of Cemiplimab for the Management of Non-Melanoma Skin Cancer: A Drug Safety Evaluation
by
Luca Potestio, Massimiliano Scalvenzi, Aimilios Lallas, Fabrizio Martora, Luigi Guerriero, Luigi Fornaro, Laura Marano and Alessia Villani
Cancers 2024, 16(9), 1732; https://doi.org/10.3390/cancers16091732 - 29 Apr 2024
Abstract
Non-melanoma skin cancer includes several types of cutaneous tumors, with basal cell carcinoma (BCC) and cutaneous squamous cell carcinoma (cSCC) as the commonest. Among the available therapeutic options, surgical excision is the mainstay of treatment for both tumors. However, tumor features and patients’
[...] Read more.
Non-melanoma skin cancer includes several types of cutaneous tumors, with basal cell carcinoma (BCC) and cutaneous squamous cell carcinoma (cSCC) as the commonest. Among the available therapeutic options, surgical excision is the mainstay of treatment for both tumors. However, tumor features and patients’ comorbidities may limit the use of these techniques, making the treatment challenging. As regards BCC, even if hedgehog inhibitors revolutionized the therapeutic scenario, there are still patients unresponsive or intolerant to these drugs. In this context, cemiplimab has been approved as second-line treatment. As regards SCC, cemiplimab was the first systemic therapy approved. The objective of this manuscript was to investigate the efficacy and safety of cemiplimab for the management of BCC and cSCC. Cemiplimab has a durable and significant effect for the management of BCC and CSCC, with a favorable safety profile. Different specialists including oncologists, radiologists, dermatologists, and surgeons are required to guarantee an integrated approach, leading to the best management of patients. Moreover, the collaboration among specialists will allow them to best manage the TEAEs, reducing the risk of treatment suspension or discontinuation. Certainly, ongoing studies and more and more emerging real-world evidence, will allow us to better characterize the role of cemiplimab for the management of advanced non-melanoma skin cancer.
Full article
(This article belongs to the Special Issue Research Progress of Cutaneous Squamous and Basal Cell Carcinomas (Volume II))
Open AccessReview
Phenotype Transformation of PitNETs
by
Zhenwei Li, Yinzi Wu, Guannan He, Renzhi Wang and Xinjie Bao
Cancers 2024, 16(9), 1731; https://doi.org/10.3390/cancers16091731 - 29 Apr 2024
Abstract
Phenotype transformation in pituitary neuroendocrine tumors is a little-known and unpredictable clinical phenomenon. Previous studies have not clearly defined and systematically concluded on the causes of this rare phenomenon. Additionally, the mechanisms of phenotype transformation are not well known. We reviewed cases reported
[...] Read more.
Phenotype transformation in pituitary neuroendocrine tumors is a little-known and unpredictable clinical phenomenon. Previous studies have not clearly defined and systematically concluded on the causes of this rare phenomenon. Additionally, the mechanisms of phenotype transformation are not well known. We reviewed cases reported in the literature with the aim of defining phenotype transformation in pituitary neuroendocrine tumors. We present an overview of the wide spectrum of phenotype transformation and its clinical features. We also discuss findings on the potential mechanism of this rare transformation, which may be related to PC1/3, the bioactivity of secretory hormones, gene mutations and the plasticity of pituitary neuroendocrine tumors. Clinicians should be aware of this rare phenomenon and more studies on the underlying mechanisms are required.
Full article
(This article belongs to the Section Clinical Research of Cancer)
►▼
Show Figures
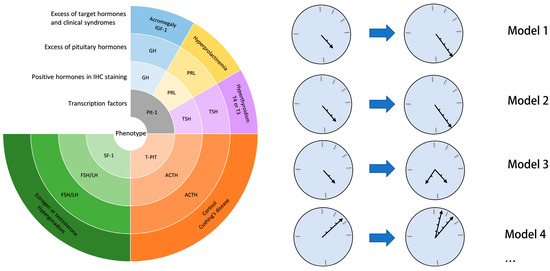
Figure 1
Open AccessArticle
Debulking Hepatectomy for Colorectal Liver Metastasis Conveys Survival Benefit
by
Jennifer A. Kalil, Lucyna Krzywon, Oran Zlotnik, Hugo Perrier, Stephanie K. Petrillo, Prosanto Chaudhury, Erik Schadde and Peter Metrakos
Cancers 2024, 16(9), 1730; https://doi.org/10.3390/cancers16091730 - 29 Apr 2024
Abstract
(1) Background: Despite advances in surgical technique and systemic chemotherapy, some patients with multifocal, bilobar colorectal liver metastases (CRLM) remain unresectable. These patients may benefit from surgical debulking of liver tumors in combination with chemotherapy compared to chemotherapy alone. (2) Methods: A retrospective
[...] Read more.
(1) Background: Despite advances in surgical technique and systemic chemotherapy, some patients with multifocal, bilobar colorectal liver metastases (CRLM) remain unresectable. These patients may benefit from surgical debulking of liver tumors in combination with chemotherapy compared to chemotherapy alone. (2) Methods: A retrospective study including patients evaluated for curative intent resection of CRLM was performed. Patients were divided into three groups: those who underwent liver resection with recurrence within 6 months (subtotal debulked, SD), those who had the first stage only of a two-stage hepatectomy (partially debulked, PD), and those never debulked (ND). Kaplan–Meier survival curves and log-rank test were performed to assess the median survival of each group. (3) Results: 174 patients underwent liver resection, and 34 patients recurred within 6 months. Of the patients planned for two-stage hepatectomy, 35 underwent the first stage only. Thirty-two patients were never resected. Median survival of the SD, PD, and ND groups was 31 months, 31 months, and 19.5 months, respectively (p = 0.012); (4) Conclusions: Patients who underwent a debulking of CRLM demonstrated a survival benefit compared to patients who did not undergo any surgical resection. This study provides support for the evaluation of intentional debulking versus palliative chemotherapy alone in a randomized trial.
Full article
(This article belongs to the Section Cancer Metastasis)
►▼
Show Figures
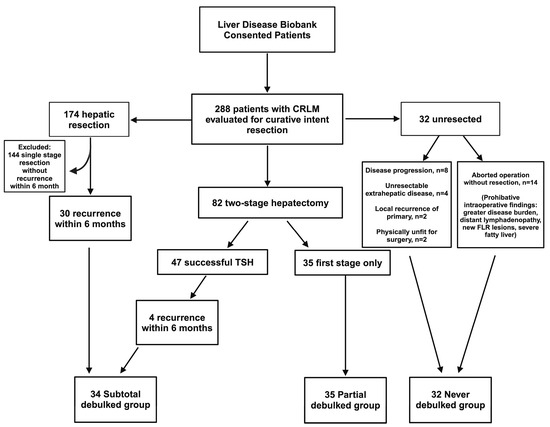
Figure 1
Open AccessReview
Hypoxia-Inducible Factor-Dependent and Independent Mechanisms Underlying Chemoresistance of Hypoxic Cancer Cells
by
Peter Wai Tik Lee, Lina Rochelle Koseki, Takao Haitani, Hiroshi Harada and Minoru Kobayashi
Cancers 2024, 16(9), 1729; https://doi.org/10.3390/cancers16091729 - 29 Apr 2024
Abstract
In hypoxic regions of malignant solid tumors, cancer cells acquire resistance to conventional therapies, such as chemotherapy and radiotherapy, causing poor prognosis in patients with cancer. It is widely recognized that some of the key genes behind this are hypoxia-inducible transcription factors, e.g.,
[...] Read more.
In hypoxic regions of malignant solid tumors, cancer cells acquire resistance to conventional therapies, such as chemotherapy and radiotherapy, causing poor prognosis in patients with cancer. It is widely recognized that some of the key genes behind this are hypoxia-inducible transcription factors, e.g., hypoxia-inducible factor 1 (HIF-1). Since HIF-1 activity is suppressed by two representative 2-oxoglutarate-dependent dioxygenases (2-OGDDs), PHDs (prolyl-4-hydroxylases), and FIH-1 (factor inhibiting hypoxia-inducible factor 1), the inactivation of 2-OGDD has been associated with cancer therapy resistance by the activation of HIF-1. Recent studies have also revealed the importance of hypoxia-responsive mechanisms independent of HIF-1 and its isoforms (collectively, HIFs). In this article, we collate the accumulated knowledge of HIF-1-dependent and independent mechanisms responsible for resistance of hypoxic cancer cells to anticancer drugs and briefly discuss the interplay between hypoxia responses, like EMT and UPR, and chemoresistance. In addition, we introduce a novel HIF-independent mechanism, which is epigenetically mediated by an acetylated histone reader protein, ATAD2, which we recently clarified.
Full article
(This article belongs to the Special Issue The Role of Hypoxia in Cancer Progression, Angiogenesis, Metastasis and Resistance to Therapy)
►▼
Show Figures
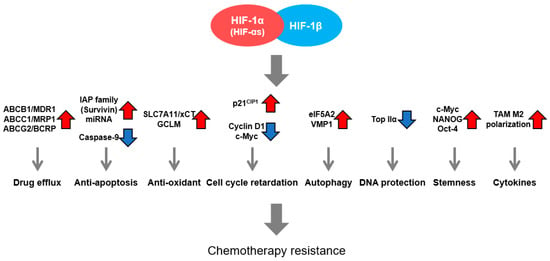
Figure 1
Open AccessSystematic Review
FDG-PET in Chimeric Antigen Receptor T-Cell (CAR T-Cell) Therapy Toxicity: A Systematic Review
by
Akram Al-Ibraheem, Ahmed Saad Abdlkadir, Egesta Lopci, Sudqi Allouzi, Diana Paez, Maryam Alkuwari, Mohammad Makoseh, Fuad Novruzov, Sharjeel Usmani, Kamal Al-Rabi and Asem Mansour
Cancers 2024, 16(9), 1728; https://doi.org/10.3390/cancers16091728 - 29 Apr 2024
Abstract
The utilization of chimeric antigen receptor (CAR) T-cell therapy to target cluster of differentiation (CD)19 in cancer immunotherapy has been a recent and significant advancement. Although this approach is highly specific and selective, it is not without complications. Therefore, a systematic review was
[...] Read more.
The utilization of chimeric antigen receptor (CAR) T-cell therapy to target cluster of differentiation (CD)19 in cancer immunotherapy has been a recent and significant advancement. Although this approach is highly specific and selective, it is not without complications. Therefore, a systematic review was conducted to assess the current state of positron emission tomography (PET) in evaluating the adverse effects induced by CAR T-cell therapy. A thorough search of relevant articles was performed in databases such as PubMed, Scopus, and Web of Science up until March 2024. Two reviewers independently selected articles and extracted data, which was then organized and categorized using Microsoft Excel. The risk of bias and methodological quality was assessed. In total, 18 articles were examined, involving a total of 753 patients, in this study. A wide range of utilities were analyzed, including predictive, correlative, and diagnostic utilities. While positive outcomes were observed in all the mentioned areas, quantitative analysis of the included studies was hindered by their heterogeneity and use of varying PET-derived parameters. This study offers a pioneering exploration of this promising field, with the goal of encouraging further and more focused research in upcoming clinical trials.
Full article
(This article belongs to the Special Issue PET/CT in Tumor Immunotherapy Assessment)
►▼
Show Figures
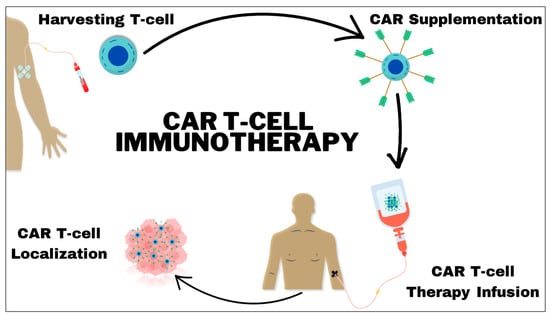
Figure 1
Open AccessReview
The Tumor Stroma of Squamous Cell Carcinoma: A Complex Environment That Fuels Cancer Progression
by
Alexandra Buruiană, Bogdan-Alexandru Gheban, Ioana-Andreea Gheban-Roșca, Carmen Georgiu, Doința Crișan and Maria Crișan
Cancers 2024, 16(9), 1727; https://doi.org/10.3390/cancers16091727 - 29 Apr 2024
Abstract
The tumor microenvironment (TME), a complex assembly of cellular and extracellular matrix (ECM) components, plays a crucial role in driving tumor progression, shaping treatment responses, and influencing metastasis. This narrative review focuses on the cutaneous squamous cell carcinoma (cSCC) tumor stroma, highlighting its
[...] Read more.
The tumor microenvironment (TME), a complex assembly of cellular and extracellular matrix (ECM) components, plays a crucial role in driving tumor progression, shaping treatment responses, and influencing metastasis. This narrative review focuses on the cutaneous squamous cell carcinoma (cSCC) tumor stroma, highlighting its key constituents and their dynamic contributions. We examine how significant changes within the cSCC ECM—specifically, alterations in fibronectin, hyaluronic acid, laminins, proteoglycans, and collagens—promote cancer progression, metastasis, and drug resistance. The cellular composition of the cSCC TME is also explored, detailing the intricate interplay of cancer-associated fibroblasts (CAFs), mesenchymal stem cells (MSCs), endothelial cells, pericytes, adipocytes, and various immune cell populations. These diverse players modulate tumor development, angiogenesis, and immune responses. Finally, we emphasize the TME’s potential as a therapeutic target. Emerging strategies discussed in this review include harnessing the immune system (adoptive cell transfer, checkpoint blockade), hindering tumor angiogenesis, disrupting CAF activity, and manipulating ECM components. These approaches underscore the vital role that deciphering TME interactions plays in advancing cSCC therapy. Further research illuminating these complex relationships will uncover new avenues for developing more effective treatments for cSCC.
Full article
(This article belongs to the Special Issue Tumor-Associated Microenvironments and Inflammation)
►▼
Show Figures
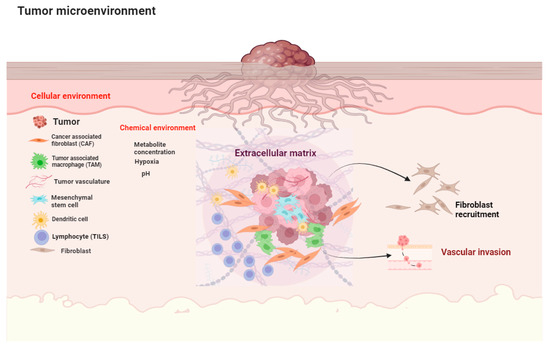
Figure 1

Journal Menu
► ▼ Journal Menu-
- Cancers Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Bioengineering, Biomolecules, Cancers, Diseases, Nanomaterials, Pharmaceutics
Dynamic Nano-Biomaterials in Tissue Regeneration and Cancer Therapies
Topic Editors: Ramar Thangam, Heemin Kang, Bibin G. Anand, Ramachandran Vijayan, Venugopal KrishnanDeadline: 15 May 2024
Topic in
Biomedicines, Cancers, JFB, Nanomaterials, Polymers
Advanced Functional Materials for Regenerative Medicine
Topic Editors: Antonino Morabito, Luca ValentiniDeadline: 6 June 2024
Topic in
Biology, Cancers, Current Oncology, Diseases, JCM, Pathogens
Pathogenetic, Diagnostic and Therapeutic Perspectives in Head and Neck Cancer
Topic Editors: Shun-Fa Yang, Ming-Hsien ChienDeadline: 20 June 2024
Topic in
Cancers, Cells, JCM, Radiation, Pharmaceutics, Applied Sciences, Nanomaterials, Current Oncology
Innovative Radiation Therapies
Topic Editors: Gérard Baldacchino, Eric Deutsch, Marie Dutreix, Sandrine Lacombe, Erika Porcel, Charlotte Robert, Emmanuelle Bourneuf, João Santos Sousa, Aurélien de la LandeDeadline: 30 June 2024

Conferences
Special Issues
Special Issue in
Cancers
Innovations in Diagnosis and Treatment of Colon and Rectal Cancer: Preoperative Optimisation, Multidisciplinary Management, and Surgical Technology Advancement
Guest Editor: Gianluca PellinoDeadline: 30 April 2024
Special Issue in
Cancers
Molecular and Cellular Heterogeneity in an Evolving Tumor Landscape: When Diversity Gives Rise to Aggressive and Drug Resistant Cells
Guest Editor: Michelle R. DawsonDeadline: 20 May 2024
Special Issue in
Cancers
Innovations in Soft Tissue Sarcoma Diagnosis and Treatment
Guest Editors: Erlinda M. Gordon, Sant P. Chawla, Frederick L. HallDeadline: 30 May 2024
Special Issue in
Cancers
Perioperative Care and Pain Management in Cancer Patients: From Basic Science to Clinical Practice
Guest Editors: Dario Bugada, Juan P. CataDeadline: 20 June 2024
Topical Collections
Topical Collection in
Cancers
Drug Resistance and Novel Therapies in Cancers
Collection Editor: Zhixiang Wang










