-
 Assessment of Some Unsymmetrical Porphyrins as Promising Molecules for Photodynamic Therapy of Cutaneous Disorders
Assessment of Some Unsymmetrical Porphyrins as Promising Molecules for Photodynamic Therapy of Cutaneous Disorders -
 Antenatal Magnesium Sulfate Benefits Female Preterm Infants but Results in Poor Male Outcomes
Antenatal Magnesium Sulfate Benefits Female Preterm Infants but Results in Poor Male Outcomes -
 Alpha-Emitting Radionuclides: Current Status and Future Perspectives
Alpha-Emitting Radionuclides: Current Status and Future Perspectives -
 A Review of Current and Pipeline Drugs for Treatment of Melanoma
A Review of Current and Pipeline Drugs for Treatment of Melanoma -
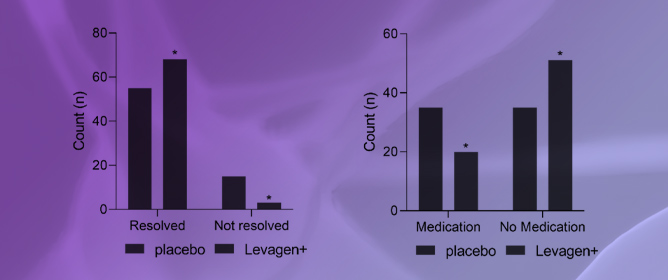 Effectiveness of Palmitoylethanolamide (Levagen+) Compared to a Placebo for Reducing Pain, Duration, and Medication Use during Migraines in Otherwise Healthy Participants—A Double-Blind Randomised Controlled Study
Effectiveness of Palmitoylethanolamide (Levagen+) Compared to a Placebo for Reducing Pain, Duration, and Medication Use during Migraines in Otherwise Healthy Participants—A Double-Blind Randomised Controlled Study
Journal Description
Pharmaceuticals
Pharmaceuticals
is a peer-reviewed, open access journal of medicinal chemistry and related drug sciences, published monthly online by MDPI. The Academy of Pharmaceutical Sciences (APS) is partners of Pharmaceuticals and their members receive a discount on the article processing charge.
- Open Access free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q2 (Pharmacology & Pharmacy) / CiteScore - Q2 (Pharmaceutical Science)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 14.6 days after submission; acceptance to publication is undertaken in 3.6 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Testimonials: See what our editors and authors say about Pharmaceuticals.
- International Electronic Conference on Medicinal Chemistry (https://sciforum.net/series/ecmc/latest)
- Companion journals for Pharmaceuticals include: Pharmacoepidemiology, Psychoactives and Drugs and Drug Candidates.
Impact Factor:
4.6 (2022);
5-Year Impact Factor:
4.9 (2022)
Latest Articles
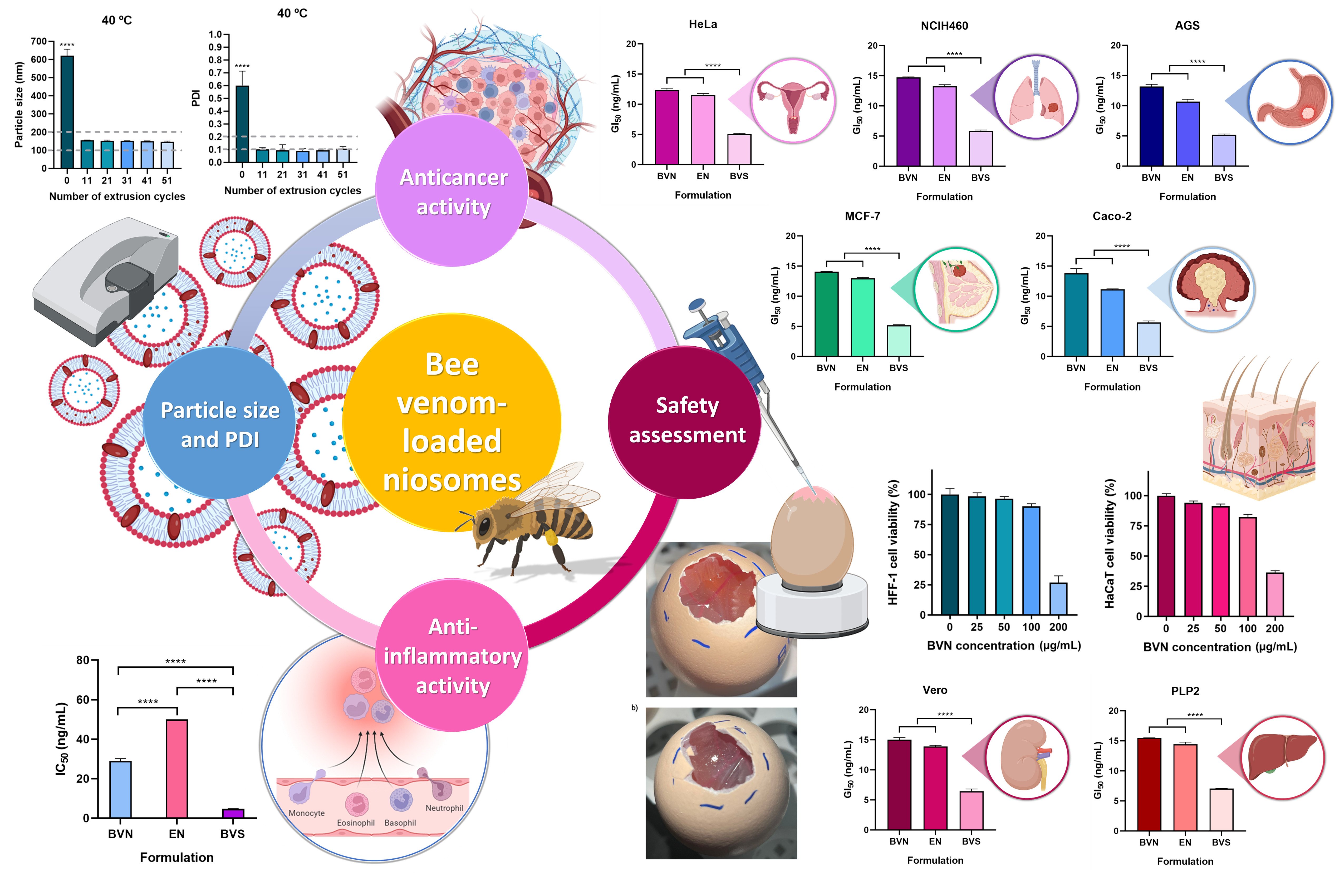
Bee Venom-Loaded Niosomes as Innovative Platforms for Cancer Treatment: Development and Therapeutical Efficacy and Safety Evaluation
Pharmaceuticals 2024, 17(5), 572; https://doi.org/10.3390/ph17050572 (registering DOI) - 29 Apr 2024
Abstract
Despite past efforts towards therapeutical innovation, cancer remains a highly incident and lethal disease, with current treatments lacking efficiency and leading to severe side effects. Hence, it is imperative to develop new, more efficient, and safer therapies. Bee venom has proven to have
[...] Read more.
Despite past efforts towards therapeutical innovation, cancer remains a highly incident and lethal disease, with current treatments lacking efficiency and leading to severe side effects. Hence, it is imperative to develop new, more efficient, and safer therapies. Bee venom has proven to have multiple and synergistic bioactivities, including antitumor effects. Nevertheless, some toxic effects have been associated with its administration. To tackle these issues, in this work, bee venom-loaded niosomes were developed, for cancer treatment. The vesicles had a small (150 nm) and homogeneous (polydispersity index of 0.162) particle size, and revealed good therapeutic efficacy in in vitro gastric, colorectal, breast, lung, and cervical cancer models (inhibitory concentrations between 12.37 ng/mL and 14.72 ng/mL). Additionally, they also revealed substantial anti-inflammatory activity (inhibitory concentration of 28.98 ng/mL), effects complementary to direct antitumor activity. Niosome safety was also assessed, both in vitro (skin, liver, and kidney cells) and ex vivo (hen’s egg chorioallantoic membrane), and results showed that compound encapsulation increased its safety. Hence, small, and homogeneous bee venom-loaded niosomes were successfully developed, with substantial anticancer and anti-inflammatory effects, making them potentially promising primary or adjuvant cancer therapies. Future research should focus on evaluating the potential of the developed platform in in vivo models.
Full article
(This article belongs to the Special Issue Pharmacological Properties and Therapeutic Potential of Honey Bee Products)
►
Show Figures
Open AccessReview
The Therapeutic Potential of Essential Oils in Managing Inflammatory Skin Conditions: A Scoping Review
by
Anouk E. W. K. Dontje, Catharina C. M. Schuiling-Veninga, Florence P. A. M. van Hunsel, Corine Ekhart, Fatih Demirci and Herman J. Woerdenbag
Pharmaceuticals 2024, 17(5), 571; https://doi.org/10.3390/ph17050571 (registering DOI) - 29 Apr 2024
Abstract
Conventional therapy is commonly used for the treatment of inflammatory skin conditions, but undesirable effects, such as erythema, dryness, skin thinning, and resistance to treatment, may cause poor patient compliance. Therefore, patients may seek complementary treatment with herbal plant products including essential oils
[...] Read more.
Conventional therapy is commonly used for the treatment of inflammatory skin conditions, but undesirable effects, such as erythema, dryness, skin thinning, and resistance to treatment, may cause poor patient compliance. Therefore, patients may seek complementary treatment with herbal plant products including essential oils (EOs). This scoping review aims to generate a broad overview of the EOs used to treat inflammatory skin conditions, namely, acne vulgaris, dermatitis and eczema, psoriasis, and rosacea, in a clinical setting. The quality, efficacy, and safety of various EOs, as well as the way in which they are prepared, are reviewed, and the potential, as well as the limitations, of EOs for the treatment of inflammatory skin conditions are discussed. Twenty-nine eligible studies (case studies, uncontrolled clinical studies, and randomized clinical studies) on the applications of EOs for inflammatory skin conditions were retrieved from scientific electronic databases (PubMed, Embase, Scopus, and the Cochrane Library). As an initial result, tea tree (Melaleuca alternifolia) oil emerged as the most studied EO. The clinical studies with tea tree oil gel for acne treatment showed an efficacy with fewer adverse reactions compared to conventional treatments. The uncontrolled studies indicated the potential efficacy of ajwain (Trachyspermum ammi) oil, eucalyptus (Eucalyptus globulus) oil, and cedarwood (Cedrus libani) oil in the treatment of acne, but further research is required to reach conclusive evidence. The placebo-controlled studies revealed the positive effects of kānuka (Kunzea ericoides) oil and frankincense (Boswellia spp.) oil in the treatment of psoriasis and eczema. The quality verification of the EO products was inconsistent, with some studies lacking analyses and transparency. The quality limitations of some studies included a small sample size, a short duration, and the absence of a control group. This present review underscores the need for extended, well-designed clinical studies to further assess the efficacy and safety of EOs for treating inflammatory skin conditions with products of assured quality and to further elucidate the mechanisms of action involved.
Full article
(This article belongs to the Special Issue Multi-Targeted Natural Products as Therapeutics)
Open AccessArticle
Synthesis and Evaluation of 5-(Heteroarylmethylene)hydantoins as Glycogen Synthase Kinase-3β Inhibitors
by
Nicholas O. Schneider, Kendra Gilreath, Daniel J. Burkett, Martin St. Maurice and William A. Donaldson
Pharmaceuticals 2024, 17(5), 570; https://doi.org/10.3390/ph17050570 (registering DOI) - 29 Apr 2024
Abstract
Glycogen synthase kinase-3 (GSK-3) is a serine/threonine kinase which plays a center role in the phosphorylation of a wide variety of proteins, generally leading to their inactivation. As such, GSK-3 is viewed as a therapeutic target. An ever-increasing number of small organic molecule
[...] Read more.
Glycogen synthase kinase-3 (GSK-3) is a serine/threonine kinase which plays a center role in the phosphorylation of a wide variety of proteins, generally leading to their inactivation. As such, GSK-3 is viewed as a therapeutic target. An ever-increasing number of small organic molecule inhibitors of GSK-3 have been reported. Phenylmethylene hydantoins are known to exhibit a wide range of inhibitory activities including for GSK-3β. A family of fourteen 2-heterocycle substituted methylene hydantoins (14, 17–29) were prepared and evaluated for the inhibition of GSK-3β at 25 μM. The IC50 values of five of these compounds was determined; the two best inhibitors are 5-[(4′-chloro-2-pyridinyl)methylene]hydantoin (IC50 = 2.14 ± 0.18 μM) and 5-[(6′-bromo-2-pyridinyl)methylene]hydantoin (IC50 = 3.39 ± 0.16 μM). The computational docking of the compounds with GSK-3β (pdb 1q41) revealed poses with hydrogen bonding to the backbone at Val135. The 5-[(heteroaryl)methylene]hydantoins did not strongly inhibit other metalloenzymes, demonstrating poor inhibitory activity against matrix metalloproteinase-12 at 25 μM and against human carbonic anhydrase at 200 μM, and were not inhibitors for Staphylococcus aureus pyruvate carboxylase at concentrations >1000 μM.
Full article
(This article belongs to the Special Issue Nitrogen Containing Scaffolds in Medicinal Chemistry 2023)
►▼
Show Figures
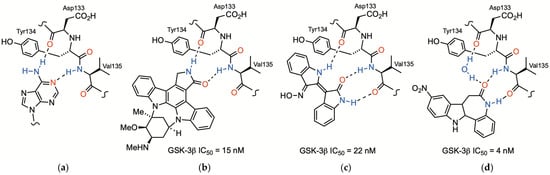
Figure 1
Open AccessArticle
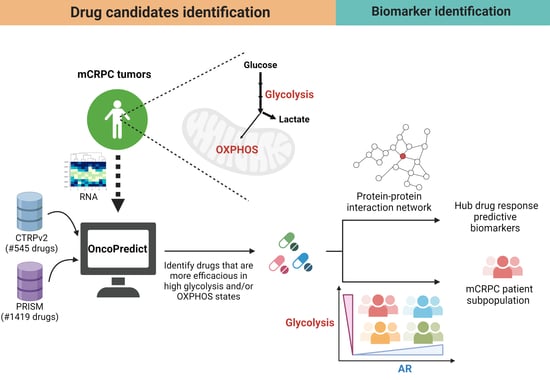
Computational Modeling to Identify Drugs Targeting Metastatic Castration-Resistant Prostate Cancer Characterized by Heightened Glycolysis
by
Mei-Chi Su, Adam M. Lee, Weijie Zhang, Danielle Maeser, Robert F. Gruener, Yibin Deng and R. Stephanie Huang
Pharmaceuticals 2024, 17(5), 569; https://doi.org/10.3390/ph17050569 (registering DOI) - 29 Apr 2024
Abstract
Metastatic castration-resistant prostate cancer (mCRPC) remains a deadly disease due to a lack of efficacious treatments. The reprogramming of cancer metabolism toward elevated glycolysis is a hallmark of mCRPC. Our goal is to identify therapeutics specifically associated with high glycolysis. Here, we established
[...] Read more.
Metastatic castration-resistant prostate cancer (mCRPC) remains a deadly disease due to a lack of efficacious treatments. The reprogramming of cancer metabolism toward elevated glycolysis is a hallmark of mCRPC. Our goal is to identify therapeutics specifically associated with high glycolysis. Here, we established a computational framework to identify new pharmacological agents for mCRPC with heightened glycolysis activity under a tumor microenvironment, followed by in vitro validation. First, using our established computational tool, OncoPredict, we imputed the likelihood of drug responses to approximately 1900 agents in each mCRPC tumor from two large clinical patient cohorts. We selected drugs with predicted sensitivity highly correlated with glycolysis scores. In total, 77 drugs predicted to be more sensitive in high glycolysis mCRPC tumors were identified. These drugs represent diverse mechanisms of action. Three of the candidates, ivermectin, CNF2024, and P276-00, were selected for subsequent vitro validation based on the highest measured drug responses associated with glycolysis/OXPHOS in pan-cancer cell lines. By decreasing the input glucose level in culture media to mimic the mCRPC tumor microenvironments, we induced a high-glycolysis condition in PC3 cells and validated the projected higher sensitivity of all three drugs under this condition (p < 0.0001 for all drugs). For biomarker discovery, ivermectin and P276-00 were predicted to be more sensitive to mCRPC tumors with low androgen receptor activities and high glycolysis activities (AR(low)Gly(high)). In addition, we integrated a protein–protein interaction network and topological methods to identify biomarkers for these drug candidates. EEF1B2 and CCNA2 were identified as key biomarkers for ivermectin and CNF2024, respectively, through multiple independent biomarker nomination pipelines. In conclusion, this study offers new efficacious therapeutics beyond traditional androgen-deprivation therapies by precisely targeting mCRPC with high glycolysis.
Full article
(This article belongs to the Special Issue Novel Therapies for the Treatment of Metastatic Prostate Cancer)
►▼
Show Figures
Graphical abstract
Open AccessReview
Classical and Novel Lipid-Lowering Therapies for Diabetic Patients with Established Coronary Artery Disease or High Risk of Coronary Artery Disease—A Narrative Clinical Review
by
Nikolaos Velidakis, Panagiotis Stachteas, Evangelia Gkougkoudi, Christodoulos Papadopoulos and Nikolaos P. E. Kadoglou
Pharmaceuticals 2024, 17(5), 568; https://doi.org/10.3390/ph17050568 (registering DOI) - 29 Apr 2024
Abstract
Diabetic atherosclerosis is a complex process that is characterized by diffuse and unstable lesions increasing 2–4-fold the risk of adverse cardiovascular (CV) events. Diabetic dyslipidemia has a predominant role in coronary artery disease (CAD) and has been the target of classical and emerging
[...] Read more.
Diabetic atherosclerosis is a complex process that is characterized by diffuse and unstable lesions increasing 2–4-fold the risk of adverse cardiovascular (CV) events. Diabetic dyslipidemia has a predominant role in coronary artery disease (CAD) and has been the target of classical and emerging pharmaceutical agents with established or promising CV benefits. The aim of the present narrative review was to summarize the effects of classical and novel lipid-lowering pharmaceutical agents on lipid profile and CV outcomes in diabetic patients with established CAD or high risk of CAD. Statins remain the first-line treatment for all diabetic patients since they considerably ameliorate lipid parameters and non-lipid CV risk factors, leading to reduced CV morbidity and mortality. Complementary to statins, ezetimibe exerts lipid-lowering properties with modest but significant reductions in major adverse cardiovascular events (MACEs) and CV mortality. PCSK9 inhibitors considerably reduce LDL-C levels and lower MACEs in diabetic patients. On the other hand, fibrates may confer a very modest decline in MACE incidence, while the CV impact of omega-3 fatty acids is promising but remains questionable. Bempedoic acid and inclisiran have a potential therapeutic role in the management of diabetic dyslipidemia, but this is still not adequately documented. Given the heightened CV risk among individuals with diabetes, more decisive results would be of great importance in the utility of all these drugs.
Full article
(This article belongs to the Section Pharmacology)
►▼
Show Figures
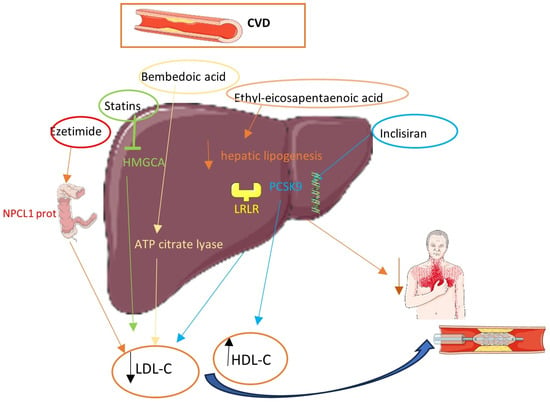
Figure 1
Open AccessArticle
Persistence in the Methadone Maintenance Program and Its Relationship with the Medication Regimen Complexity Index in Opioid-Dependent Patients
by
Elena Alba Álvaro-Alonso, María del Carmen Gómez-Álvarez, Beatriz Segovia-Tapiador, María Isabel Del-Pino-Illaconza, Jorge Valencia, Pablo Ryan, Antonio Aguilar-Ros and Ismael Escobar-Rodríguez
Pharmaceuticals 2024, 17(5), 567; https://doi.org/10.3390/ph17050567 (registering DOI) - 29 Apr 2024
Abstract
It has been shown that the Medication Regimen Complexity Index (MRCI) is a useful and reliable tool for calculating the complexity of the pharmacotherapeutic regimen (CPR). Furthermore, a high MRCI is associated with lower adherence. However, the MRCI of opioid-dependent patients (ODP) has
[...] Read more.
It has been shown that the Medication Regimen Complexity Index (MRCI) is a useful and reliable tool for calculating the complexity of the pharmacotherapeutic regimen (CPR). Furthermore, a high MRCI is associated with lower adherence. However, the MRCI of opioid-dependent patients (ODP) has not been studied. The aim of this study is to calculate the Methadone Maintenance Program (MMP) persistence and the MRCI score in a ODP cohort. Second, to analyze its relationship and association with other variables. To accomplish this research, an observational study including adults with a confirmed diagnosis of opiate-dependency according to the DSM-5 in a MMP center was carried out. To define MMP-persistence, a group was created by the researchers who defined five weighted items according to their agreed importance. Our first contribution was to create a new definition of MMP-persistence. This study also identified age, comorbidities, and received methadone maintenance doses as successful predictors for MMP-persistence. We have also shown that the MRCI does not seem to be a useful tool to determine MMP-persistence, probably because there are multiple factors that influence it in addition to the CPR. It is necessary to continue searching for more precise selection and stratification tools for ODP to improve their persistence.
Full article
(This article belongs to the Special Issue Drug Safety and Relevant Issues in the Real-World 2024)
Open AccessArticle
Effect of Lampaya medicinalis Phil. (Verbenaceae) and Palmitic Acid on Insulin Signaling and Inflammatory Marker Expression in Human Adipocytes
by
Gabriela Yuri, Mariana Cifuentes, Pedro Cisternas, Adrián Paredes and Paulina Ormazabal
Pharmaceuticals 2024, 17(5), 566; https://doi.org/10.3390/ph17050566 (registering DOI) - 29 Apr 2024
Abstract
Background: Aging and obesity are associated with insulin resistance (IR) and low-grade inflammation. Molecularly, IR is characterized by a reduction in glucose uptake and insulin signaling (IRS-1/Akt/AS160 pathway), while inflammation may result from upregulated NF-κB pathway after low Tyr-IκBα phosphorylation. Upregulated phosphatase activity
[...] Read more.
Background: Aging and obesity are associated with insulin resistance (IR) and low-grade inflammation. Molecularly, IR is characterized by a reduction in glucose uptake and insulin signaling (IRS-1/Akt/AS160 pathway), while inflammation may result from upregulated NF-κB pathway after low Tyr-IκBα phosphorylation. Upregulated phosphatase activity of PTP1B is associated with impaired insulin signaling and increased inflammation. Plasma levels of palmitic acid (PA) are elevated in obesity, triggering inflammation and disruption of insulin signaling. Traditional medicine in Northern Chile uses oral infusions of Lampaya medicinalis Phil. (Verbenaceae) to treat inflammatory conditions. Significant amounts of flavonoids are found in the hydroethanolic extract of Lampaya (HEL), which may account for its biological activity. The aim of this work was to study the effect of HEL and PA on insulin signaling and glucose uptake as well as inflammatory marker expression in human adipocytes. Methods: We studied HEL effects on PA-induced impairment on insulin signaling, glucose uptake and inflammatory marker content in human SW872 adipocytes. HEL cytotoxicity was assessed in adipocytes at different concentrations (0.01 to 10 g/mL). Adipocytes were incubated or not with PA (0.4 mM, 24 h) with or without HEL (2 h pre-incubation), and then stimulated with insulin (10 min, 100 mM) or a vehicle. Phospho-IRS-1, phospho-Akt, phospho-AS160, phospho-NF-κB and phospho-IκBα, as well as protein levels of PTP1B, were assessed using Western blotting, and glucose uptake was evaluated using the 2-NBDG analogue. Results: At the assessed HEL concentrations, no cytotoxic effects were observed. PA decreased insulin-stimulated phospho-Akt and glucose uptake, while co-treatment with HEL increased such markers. PA decreased phospho-IRS-1 and phospho-Tyr-IκBα. On the other hand, incubation with HEL+PA decreased phospho-AS160 and phospho-NF-κB compared with cells treated with PA alone. Conclusion: Our results suggest a beneficial effect of HEL by improving PA-induced impairment on molecular markers of insulin signaling, glucose uptake and inflammation in adipocytes. Further studies are necessary to elucidate whether lampaya may constitute a preventive strategy for people whose circulating PA levels contribute to IR and inflammation during aging and obesity.
Full article
(This article belongs to the Special Issue Medicinal Plants and Natural Compounds for Potential Use in Aging-Related Disorders)
►▼
Show Figures
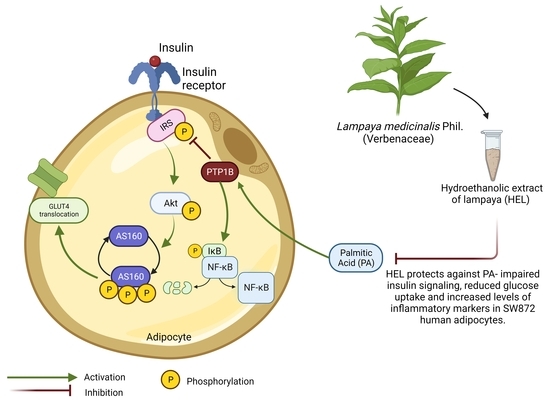
Graphical abstract
Open AccessArticle
Comprehensive Analysis of Drug Utilization Patterns, Gender Disparities, Lifestyle Influences, and Genetic Factors: Insights from Elderly Cohort Using g-Nomic® Software
by
Bárbara Rodríguez Castillo, Marc Cendrós, Carlos J. Ciudad and Ana Sabater
Pharmaceuticals 2024, 17(5), 565; https://doi.org/10.3390/ph17050565 (registering DOI) - 28 Apr 2024
Abstract
Polypharmacy is a global healthcare concern, especially among the elderly, leading to drug interactions and adverse reactions, which are significant causes of death in developed nations. However, the integration of pharmacogenetics can help mitigate these risks. In this study, the data from 483
[...] Read more.
Polypharmacy is a global healthcare concern, especially among the elderly, leading to drug interactions and adverse reactions, which are significant causes of death in developed nations. However, the integration of pharmacogenetics can help mitigate these risks. In this study, the data from 483 patients, primarily elderly and polymedicated, were analyzed using Eugenomic®’s personalized prescription software, g-Nomic®. The most prescribed drug classes included antihypertensives, platelet aggregation inhibitors, cholesterol-lowering drugs, and gastroprotective medications. Drug–lifestyle interactions primarily involved inhibitions but also included inductions. Interactions were analyzed considering gender. Significant genetic variants identified in the study encompassed ABCB1, SLCO1B1, CYP2C19, CYP2C9, CYP2D6, CYP3A4, ABCG2, NAT2, SLC22A1, and G6PD. To prevent adverse reactions and enhance medication effectiveness, it is strongly recommended to consider pharmacogenetics testing. This approach shows great promise in optimizing medication regimens and ultimately improving patient outcomes.
Full article
(This article belongs to the Special Issue Drug Safety and Relevant Issues in the Real-World 2024)
►▼
Show Figures

Figure 1
Open AccessArticle
A Comprehensive Description of the Anatomy and Histochemistry of Psychotria capillacea (Müll. Arg.) Standl. and an Investigation into Its Anti-Inflammatory Effects in Mice and Role in Scopolamine-Induced Memory Impairment
by
Anelise Samara Nazari Formagio, Wagner Vilegas, Cândida Aparecida Leite Kassuya, Valter Paes De Almeida, Jane Manfron, Elisabete Castelon Konkiewitz, Edward Benjamin Ziff, Janaine Alberto Marangoni Faoro, Jessica Maurino Dos Santos, Ana Julia Cecatto, Maria Helena Sarragiotto and Rosilda Mara Mussury
Pharmaceuticals 2024, 17(5), 564; https://doi.org/10.3390/ph17050564 (registering DOI) - 28 Apr 2024
Abstract
Species of the genus Psychotria are used in popular medicine for pain, inflammatory symptoms, and mental disorders. Psychotria capillacea (Müll. Arg.) Standl. (Rubiaceae) is commonly known as coffee and some scientific studies have demonstrated its therapeutic potential. The goal of this study was
[...] Read more.
Species of the genus Psychotria are used in popular medicine for pain, inflammatory symptoms, and mental disorders. Psychotria capillacea (Müll. Arg.) Standl. (Rubiaceae) is commonly known as coffee and some scientific studies have demonstrated its therapeutic potential. The goal of this study was to investigate the anti-inflammatory and neuroprotective effects, and acetylcholinesterase (AChE) inhibitory activity of a methanolic extract obtained from leaves of P. capillacea (MEPC), as well as the micromorphology and histochemistry of the leaves and stems of this plant. In addition, the MEPC was analyzed by UHPLC-MS/MS and the alkaloidal fraction (AF) obtained from the MEPC was tested in a mouse model of inflammation. MEPC contained three indole alkaloids, one sesquiterpene (megastigmane-type) and two terpene lactones. MEPC (3, 30 and 100 mg/kg) and AF (3 and 30 mg/kg) were evaluated in inflammation models and significantly inhibited edema at 2 h and 4 h, mechanical hyperalgesia after 4 h and the response to cold 3 h and 4 h after carrageenan injection. Scopolamine significantly increased the escape latency, and reduced the swimming time and number of crossings in the target quadrant and distance, while MEPC (3, 30 and 100 mg/kg), due to its neuroprotective actions, reversed these effects. AChE activity was significantly decreased in the cerebral cortex (52 ± 3%) and hippocampus (60 ± 3%), after MEPC administration. Moreover, micromorphological and histochemical information was presented, to aid in species identification and quality control of P. capillacea. The results of this study demonstrated that P. capillacea is an anti-inflammatory and antihyperalgesic agent that can treat acute disease and enhance memory functions in mouse models.
Full article
(This article belongs to the Section Natural Products)
►▼
Show Figures
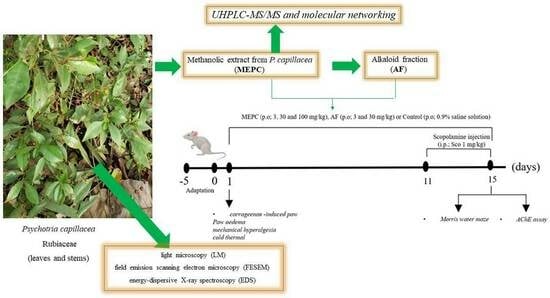
Graphical abstract
Open AccessEditorial
Methyl-Containing Pharmaceuticals
by
Davide Illuminati and Anna Fantinati
Pharmaceuticals 2024, 17(5), 563; https://doi.org/10.3390/ph17050563 (registering DOI) - 28 Apr 2024
Abstract
This Special Issue, which focused on methyl-containing pharmaceuticals, collected different papers and reviews on this topic [...]
Full article
(This article belongs to the Special Issue Methyl-Containing Pharmaceuticals)
Open AccessArticle
An Assessment of Different Decision Support Software from the Perspective of Potential Drug–Drug Interactions in Patients with Chronic Kidney Diseases
by
Muhammed Yunus Bektay, Aysun Buker Cakir, Meltem Gursu, Rumeyza Kazancioglu and Fikret Vehbi Izzettin
Pharmaceuticals 2024, 17(5), 562; https://doi.org/10.3390/ph17050562 (registering DOI) - 28 Apr 2024
Abstract
Chronic kidney disease (CKD) is a multifaceted disorder influenced by various factors. Drug–drug interactions (DDIs) present a notable risk factor for hospitalization among patients with CKD. This study aimed to assess the frequency and attributes of potential DDIs (pDDIs) in patients with CKD
[...] Read more.
Chronic kidney disease (CKD) is a multifaceted disorder influenced by various factors. Drug–drug interactions (DDIs) present a notable risk factor for hospitalization among patients with CKD. This study aimed to assess the frequency and attributes of potential DDIs (pDDIs) in patients with CKD and to ascertain the concordance among different Clinical Decision Support Software (CDSS). A cross-sectional study was conducted in a nephrology outpatient clinic at a university hospital. The pDDIs were identified and evaluated using Lexicomp® and Medscape®. The patients’ characteristics, comorbidities, and medicines used were recorded. The concordance of different CDSS were evaluated using the Kendall W coefficient. An evaluation of 1121 prescribed medications for 137 patients was carried out. The mean age of the patients was 64.80 ± 14.59 years, and 41.60% of them were male. The average year with CKD was 6.48 ± 5.66. The mean number of comorbidities was 2.28 ± 1.14. The most common comorbidities were hypertension, diabetes, and coronary artery disease. According to Medscape, 679 pDDIs were identified; 1 of them was contraindicated (0.14%), 28 (4.12%) were serious-use alternative, and 650 (9.72%) were interventions that required closely monitoring. According to Lexicomp, there were 604 drug–drug interactions. Of these interactions, 9 (1.49%) were in the X category, 60 (9.93%) were in the D category, and 535 (88.57%) were in the C category. Two different CDSS systems exhibited statistically significant concordance with poor agreement (W = 0.073, p < 0.001). Different CDSS systems are commonly used in clinical practice to detect pDDIs. However, various factors such as the operating principles of these programs and patient characteristics can lead to incorrect guidance in clinical decision making. Therefore, instead of solely relying on programs with lower reliability and consistency scores, multidisciplinary healthcare teams, including clinical pharmacists, should take an active role in identifying and preventing pDDIs.
Full article
(This article belongs to the Special Issue Pharmacovigilance in Drug Therapy: Drug–Drug Interactions and Safety Evaluation)
►▼
Show Figures
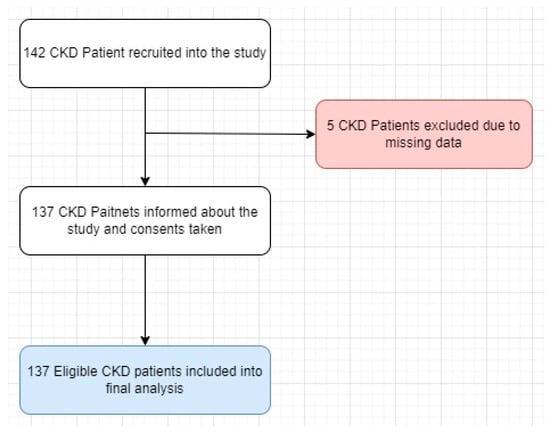
Figure 1
Open AccessReview
From Eye Care to Hair Growth: Bimatoprost
by
Marco Zeppieri, Caterina Gagliano, Leopoldo Spadea, Carlo Salati, Ekele Caleb Chukwuyem, Ehimare Samuel Enaholo, Fabiana D’Esposito and Mutali Musa
Pharmaceuticals 2024, 17(5), 561; https://doi.org/10.3390/ph17050561 (registering DOI) - 27 Apr 2024
Abstract
Background: Bimatoprost has emerged as a significant medication in the field of medicine over the past several decades, with diverse applications in ophthalmology, dermatology, and beyond. Originally developed as an ocular hypotensive agent, it has proven highly effective in treating glaucoma and ocular
[...] Read more.
Background: Bimatoprost has emerged as a significant medication in the field of medicine over the past several decades, with diverse applications in ophthalmology, dermatology, and beyond. Originally developed as an ocular hypotensive agent, it has proven highly effective in treating glaucoma and ocular hypertension. Its ability to reduce intraocular pressure has established it as a first-line treatment option, improving management and preventing vision loss. In dermatology, bimatoprost has shown promising results in the promotion of hair growth, particularly in the treatment of alopecia and hypotrichosis. Its mechanism of action, stimulating the hair cycle and prolonging the growth phase, has led to the development of bimatoprost-containing solutions for enhancing eyelash growth. Aim: The aim of our review is to provide a brief description, overview, and studies in the current literature regarding the versatile clinical use of bimatoprost in recent years. This can help clinicians determine the most suitable individualized therapy to meet the needs of each patient. Methods: Our methods involve a comprehensive review of the latest advancements reported in the literature in bimatoprost formulations, which range from traditional eye drops to sustained-release implants. These innovations offer extended drug delivery, enhance patient compliance, and minimize side effects. Results: The vast literature published on PubMed has confirmed the clinical usefulness of bimatoprost in lowering intraocular pressure and in managing patients with glaucoma. Numerous studies have shown promising results in dermatology and esthetics in promoting hair growth, particularly in treating alopecia and hypotrichosis. Its mechanism of action involves stimulating the hair cycle and prolonging the growth phase, leading to the development of solutions that enhance eyelash growth. The global use of bimatoprost has expanded significantly, with applications growing beyond its initial indications. Ongoing research is exploring its potential in glaucoma surgery, neuroprotection, and cosmetic procedures. Conclusions: Bimatoprost has shown immense potential for addressing a wide range of therapeutic needs through various formulations and advancements. Promising future perspectives include the exploration of novel delivery systems such as contact lenses and microneedles to further enhance drug efficacy and patient comfort. Ongoing research and future perspectives continue to shape its role in medicine, promising further advancements and improved patient outcomes.
Full article
(This article belongs to the Special Issue Ophthalmic Pharmacology)
►▼
Show Figures
Figure 1
Open AccessArticle
Antimicrobial Effect of Honey Phenolic Compounds against E. coli—An In Vitro Study
by
Laura Kassym, Assiya Kussainova, Yuliya Semenova and Pauline McLoone
Pharmaceuticals 2024, 17(5), 560; https://doi.org/10.3390/ph17050560 (registering DOI) - 27 Apr 2024
Abstract
Growing concern over antimicrobial resistance in chronic wound patients necessitates the exploration of alternative treatments from natural sources. This study suggests that honey’s phenolic compounds may offer antimicrobial benefits, warranting further investigation for therapeutic development. The main aim of this study was to
[...] Read more.
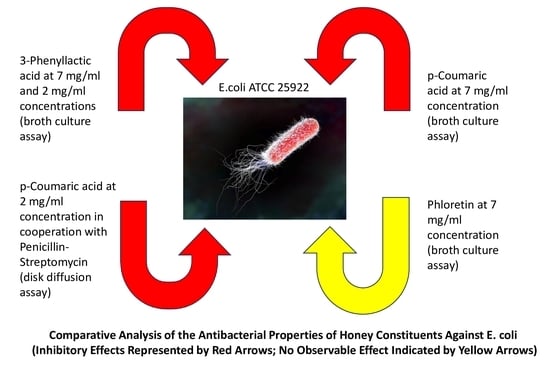
Growing concern over antimicrobial resistance in chronic wound patients necessitates the exploration of alternative treatments from natural sources. This study suggests that honey’s phenolic compounds may offer antimicrobial benefits, warranting further investigation for therapeutic development. The main aim of this study was to investigate the antimicrobial activity of phenolic compounds and to determine the effects of their sub-inhibitory concentrations against Escherichia coli (E. coli). 3-phenyllactic acid (PLA), p-coumaric acid (PCA), and phloretin were tested against the bacterial strain of E. coli ATCC 25922. Comparison of the antimicrobial activity of honey constituents in vitro was performed using a broth culture assay. Measurement of the inhibitory properties of constituents in vitro was conducted using disc and well diffusion assays. The effects of sub-inhibitory concentrations of PCA on the susceptibility of E. coli ATCC 25922 to penicillin–streptomycin were tested. The results demonstrated that PLA was the most efficient antimicrobial agent, followed by PCA, whereas phloretin, at lower (2 mg/mL) concentrations, led to an increase in the growth of E. coli. Various modifications of the agar diffusion assay did not reveal the antibacterial properties of the studied phytochemicals. The enhancing effect of a sub-inhibitory concentration of PCA in cooperation with penicillin–streptomycin was shown. These findings might be helpful for the further investigation and development of new antimicrobial agents for the treatment of skin infections and wounds.
Full article
(This article belongs to the Section Natural Products)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Development of New Drugs to Treat Tuberculosis Based on the Dinitrobenzamide Scaffold
by
Tiago Delgado, João P. Pais, David Pires, Filipe G. A. Estrada, Rita C. Guedes, Elsa Anes and Luis Constantino
Pharmaceuticals 2024, 17(5), 559; https://doi.org/10.3390/ph17050559 (registering DOI) - 27 Apr 2024
Abstract
Tuberculosis (TB) continues to be a major global health challenge and a leading cause of death from infectious diseases. Inspired by the results from a previous work by our group on antimycobacterial N-alkylnitrobenzamides, which are structurally related to the nitrobenzamide family of
[...] Read more.
Tuberculosis (TB) continues to be a major global health challenge and a leading cause of death from infectious diseases. Inspired by the results from a previous work by our group on antimycobacterial N-alkylnitrobenzamides, which are structurally related to the nitrobenzamide family of decaprenylphosphoryl-β-d-ribose oxidase (DprE1) inhibitors, the present study explored a broad array of substituted benzamides. We particularly focused on previously unexplored 3,5-dinitrobenzamide derivatives. Starting with 3,5-dinitrobenzoic acid, we synthesized a diverse library of amides, incorporating both linear and cyclic amine moieties and also assessed the impact of terminal aromatic groups connected through ether, ester, or amide bonds on the bioactivity of the compounds. The synthesis primarily utilized nucleophilic addition/elimination, SN2, and Mitsunobu reactions. The activity was impacted mainly by two structural features, the addition of an aromatic moiety as a terminal group and the type of linker. The most interesting compounds (c2, d1, and d2, MIC = 0.031 μg/mL) exhibited activities against Mycobacterium Tuberculosis (Mtb) H37Rv comparable to isoniazid. Complementary computational studies helped elucidate potential interactions with DprE1, enhancing our understanding of the molecular basis of their action. Our findings suggest that the most active compounds provide a promising foundation for the continued development of new antimycobacterial agents.
Full article
(This article belongs to the Special Issue Selected Papers from the 9th International Electronic Conference on Medicinal Chemistry (ECMC2023))
►▼
Show Figures
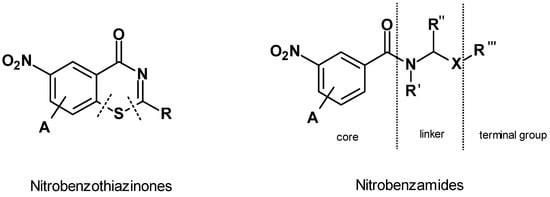
Figure 1
Open AccessArticle
Unveiling the Role of Tryptophan 2,3-Dioxygenase in the Angiogenic Process
by
Marta Cecchi, Cecilia Anceschi, Angela Silvano, Maria Luisa Coniglio, Aurora Chinnici, Lucia Magnelli, Andrea Lapucci, Anna Laurenzana and Astrid Parenti
Pharmaceuticals 2024, 17(5), 558; https://doi.org/10.3390/ph17050558 (registering DOI) - 27 Apr 2024
Abstract
Background: Indoleamine 2,3-dioxygenase (IDO1) and tryptophan-2,3-dioxygenase (TDO) are the two principals enzymes involved in the catabolization of tryptophan (Trp) into kynurenine (Kyn). Despite their well-established role in the immune escape, their involvement in angiogenesis remains uncertain. We aimed to characterize TDO and IDO1
[...] Read more.
Background: Indoleamine 2,3-dioxygenase (IDO1) and tryptophan-2,3-dioxygenase (TDO) are the two principals enzymes involved in the catabolization of tryptophan (Trp) into kynurenine (Kyn). Despite their well-established role in the immune escape, their involvement in angiogenesis remains uncertain. We aimed to characterize TDO and IDO1 in human umbilical venular endothelial cells (HUVECs) and human endothelial colony-forming cells (ECFCs). Methods: qRT-PCR and immunofluorescence were used for TDO and IDO1 expression while their activity was measured using ELISA assays. Cell proliferation was examined via MTT tests and in in vitro angiogenesis by capillary morphogenesis. Results: HUVECs and ECFCs expressed TDO and IDO1. Treatment with the selective TDO inhibitor 680C91 significantly impaired HUVEC proliferation and 3D-tube formation in response to VEGF-A, while IDO1 inhibition showed no effect. VEGF-induced mTor phosphorylation and Kyn production were hindered by 680C91. ECFC morphogenesis was also inhibited by 680C91. Co-culturing HUVECs with A375 induced TDO up-regulation in both cell types, whose inhibition reduced MMP9 activity and prevented c-Myc and E2f1 upregulation. Conclusions: HUVECs and ECFCs express the key enzymes of the kynurenine pathway. Significantly, TDO emerges as a pivotal player in in vitro proliferation and capillary morphogenesis, suggesting a potential pathophysiological role in angiogenesis beyond its well-known immunomodulatory effects.
Full article
(This article belongs to the Special Issue Pharmacological Treatments for Melanoma)
Open AccessReview
Phytosterols and the Digestive System: A Review Study from Insights into Their Potential Health Benefits and Safety
by
Edyta Miszczuk, Andrzej Bajguz, Łukasz Kiraga, Kijan Crowley and Magdalena Chłopecka
Pharmaceuticals 2024, 17(5), 557; https://doi.org/10.3390/ph17050557 (registering DOI) - 26 Apr 2024
Abstract
Phytosterols are a large group of substances belonging to sterols—compounds naturally occurring in the tissues of plants, animals, and humans. The most well-known animal sterol is cholesterol. Among phytosterols, the most significant compounds are β-sitosterol, stigmasterol, and campesterol. At present, they are mainly
[...] Read more.
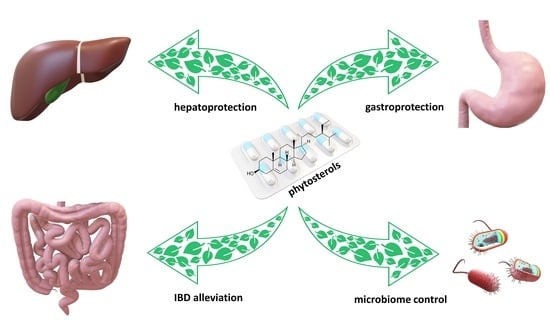
Phytosterols are a large group of substances belonging to sterols—compounds naturally occurring in the tissues of plants, animals, and humans. The most well-known animal sterol is cholesterol. Among phytosterols, the most significant compounds are β-sitosterol, stigmasterol, and campesterol. At present, they are mainly employed in functional food products designed to counteract cardiovascular disorders by lowering levels of ‘bad’ cholesterol, which stands as their most extensively studied purpose. It is currently understood that phytosterols may also alleviate conditions associated with the gastrointestinal system. Their beneficial pharmacological properties in relation to gastrointestinal tract include anti-inflammatory and hepatoprotective activity. Also, the anti-cancer properties as well as the impact on the gut microbiome could be a very interesting area of research, which might potentially lead to the discovery of their new application. This article provides consolidated knowledge on a new potential use of phytosterols, namely the treatment or prevention of gastrointestinal diseases. The cited studies indicate high therapeutic efficacy in conditions such as peptic ulcer disease, IBD or liver failure caused by hepatotoxic xenobiotics, however, these are mainly in vitro or in vivo studies. Nevertheless, studies to date indicate their therapeutic potential as adjunctive treatments to conventional therapies, which often exhibit unsatisfactory efficacy or serious side effects. Unfortunately, at this point there is a lack of significant clinical study data to use phytosterols in clinical practice in this area.
Full article
(This article belongs to the Special Issue Botanicals: Bioactive Molecules and Therapeutic Properties for Human Health 2023)
►▼
Show Figures
Graphical abstract
Open AccessCorrection
Correction: Shakir et al. Exorbitant Drug Loading of Metformin and Sitagliptin in Mucoadhesive Buccal Tablet: In Vitro and In Vivo Characterization in Healthy Volunteers. Pharmaceuticals 2022, 15, 686
by
Rouheena Shakir, Sana Hanif, Ahmad Salawi, Rabia Arshad, Rai Muhammad Sarfraz, Muhammad Irfan, Syed Atif Raza, Kashif Barkat, Fahad Y. Sabei, Yosif Almoshari, Meshal Alshamrani and Muhammad Ali Syed
Pharmaceuticals 2024, 17(5), 556; https://doi.org/10.3390/ph17050556 - 26 Apr 2024
Abstract
►▼
Show Figures
In the original publication, a mistake was observed in Figure 5 as published [...]
Full article
Figure 5
Open AccessCommunication
Switching from Beraprost to Selexipag in the Treatment of Pulmonary Arterial Hypertension: Insights from a Phase IV Study of the Japanese Registry (The EXCEL Study: EXChange from bEraprost to seLexipag Study)
by
Yuichi Tamura, Hiraku Kumamaru, Ichizo Tsujino, Rika Suda, Kohtaro Abe, Takumi Inami, Koshin Horimoto, Shiro Adachi, Satoshi Yasuda, Fusako Sera, Yu Taniguchi, Masataka Kuwana and Koichiro Tatsumi
Pharmaceuticals 2024, 17(5), 555; https://doi.org/10.3390/ph17050555 - 26 Apr 2024
Abstract
Pulmonary arterial hypertension (PAH) remains a significant challenge in cardiology, necessitating advancements in treatment strategies. This study explores the safety and efficacy of transitioning patients from beraprost to selexipag, a novel selective prostacyclin receptor agonist, within a Japanese cohort. Employing a multicenter, open-label,
[...] Read more.
Pulmonary arterial hypertension (PAH) remains a significant challenge in cardiology, necessitating advancements in treatment strategies. This study explores the safety and efficacy of transitioning patients from beraprost to selexipag, a novel selective prostacyclin receptor agonist, within a Japanese cohort. Employing a multicenter, open-label, prospective design, 25 PAH patients inadequately managed on beraprost were switched to selexipag. Key inclusion criteria included ongoing beraprost therapy for ≥3 months, a diagnosis of PAH confirmed by mean pulmonary artery pressure (mPAP) ≥ 25 mmHg, and current treatment with endothelin receptor antagonists and/or phosphodiesterase type 5 inhibitors. Outcomes assessed were changes in hemodynamic parameters (mPAP, cardiac index, pulmonary vascular resistance) and the 6 min walk distance (6-MWD) over 3–6 months. The study found no statistically significant changes in these parameters post-switch. However, a subset of patients, defined as responders, demonstrated improvements in all measured hemodynamic parameters, suggesting a potential benefit in carefully selected patients. The transition was generally well-tolerated with no serious adverse events reported. This investigation underscores the importance of personalized treatment strategies in PAH, highlighting that certain patients may benefit from switching to selexipag, particularly those previously on higher doses of beraprost. Further research is needed to elucidate the predictors of positive response to selexipag and optimize treatment regimens for this complex condition.
Full article
(This article belongs to the Special Issue The 20th Anniversary of Pharmaceuticals—Advances in Medicinal Chemistry)
►▼
Show Figures
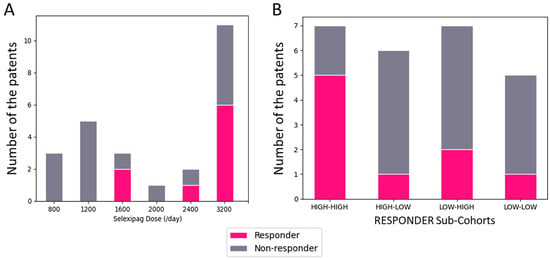
Figure 1
Open AccessArticle
Global Health Priority Box: Discovering Flucofuron as a Promising Antikinetoplastid Compound
by
Carlos J. Bethencourt-Estrella, Atteneri López-Arencibia, Jacob Lorenzo-Morales and José E. Piñero
Pharmaceuticals 2024, 17(5), 554; https://doi.org/10.3390/ph17050554 (registering DOI) - 25 Apr 2024
Abstract
Leishmaniasis, produced by Leishmania spp., and Chagas disease, produced by Trypanosoma cruzi, affect millions of people around the world. The treatments for these pathologies are not entirely effective and produce some side effects. For these reasons, it is necessary to develop new
[...] Read more.
Leishmaniasis, produced by Leishmania spp., and Chagas disease, produced by Trypanosoma cruzi, affect millions of people around the world. The treatments for these pathologies are not entirely effective and produce some side effects. For these reasons, it is necessary to develop new therapies that are more active and less toxic for patients. Some initiatives, such as the one carried out by the Medicines for Malaria Venture, allow for the screening of a large number of compounds of different origins to find alternatives to the lack of trypanocide treatments. In this work, 240 compounds were tested from the Global Health Priority Box (80 compounds with confirmed activity against drug-resistant malaria, 80 compounds for screening against neglected and zoonotic diseases and diseases at risk of drug resistance, and 80 compounds with activity against various vector species) against Trypanosoma cruzi and Leishmania amazonensis. Flucofuron, a compound with activity against vectors and with previous activity reported against Staphylococcus spp. and Schistosoma spp., demonstrates activity against L. amazonensis and T. cruzi and produces programmed cell death in the parasites. Flucofuron seems to be a good candidate for continuing study and proving its use as a trypanocidal agent.
Full article
(This article belongs to the Special Issue Antiparasitics 2023)
►▼
Show Figures
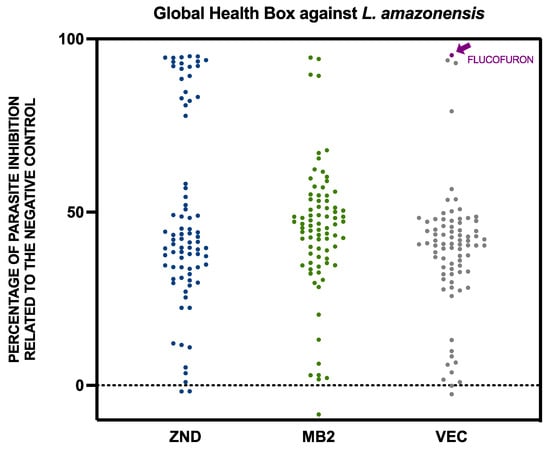
Figure 1
Open AccessArticle
Effects of CYP3A5 Genotype on Tacrolimus Pharmacokinetics and Graft-versus-Host Disease Incidence in Allogeneic Hematopoietic Stem Cell Transplantation
by
Daniel N. Marco, Mònica Molina, Ana-María Guio, Judit Julian, Virginia Fortuna, Virginia-Lucila Fabregat-Zaragoza, María-Queralt Salas, Inés Monge-Escartín, Gisela Riu-Viladoms, Esther Carcelero, Joan Ramón Roma, Noemí Llobet, Jordi Arcarons, María Suárez-Lledó, Laura Rosiñol, Francesc Fernández-Avilés, Montserrat Rovira, Mercè Brunet and Carmen Martínez
Pharmaceuticals 2024, 17(5), 553; https://doi.org/10.3390/ph17050553 - 25 Apr 2024
Abstract
Tacrolimus (Tac) is pivotal in preventing acute graft-versus-host disease (GVHD) after allogeneic hematopoietic stem cell transplantation (alloHSCT). It has been reported that genetic factors, including CYP3A5*3 and CYP3A4*22 polymorphisms, have an impact on Tac metabolism, dose requirement, and response to
[...] Read more.
Tacrolimus (Tac) is pivotal in preventing acute graft-versus-host disease (GVHD) after allogeneic hematopoietic stem cell transplantation (alloHSCT). It has been reported that genetic factors, including CYP3A5*3 and CYP3A4*22 polymorphisms, have an impact on Tac metabolism, dose requirement, and response to Tac. There is limited information regarding this topic in alloHSCT. The CYP3A5 genotype and a low Tac trough concentration/dose ratio (Tac C0/D ratio) can be used to identify fast metabolizers and predict the required Tac dose to achieve target concentrations earlier. We examined 62 Caucasian alloHSCT recipients with a fast metabolizer phenotype (C0/dose ratio ≤ 1.5 ng/mL/mg), assessing CYP3A5 genotypes and acute GVHD incidence. Forty-nine patients (79%) were poor metabolizers (2 copies of the variant *3 allele) and 13 (21%) were CYP3A5 expressers (CYP3A5*1/*1 or CYP3A5*1/*3 genotypes). CYP3A5 expressers had lower C0 at 48 h (3.7 vs. 6.2 ng/mL, p = 0.03) and at 7 days (8.6 vs. 11.4 ng/mL, p = 0.04) after Tac initiation, tended to take longer to reach Tac therapeutic range (11.8 vs. 8.9 days, p = 0.16), and had higher incidence of both global (92.3% vs. 38.8%, p < 0.001) and grade II-IV acute GVHD (61.5% vs. 24.5%, p = 0.008). These results support the adoption of preemptive pharmacogenetic testing to better predict individual Tac initial dose, helping to achieve the therapeutic range and reducing the risk of acute GVHD earlier.
Full article
(This article belongs to the Special Issue Therapeutic Drug Monitoring and Adverse Drug Reactions)
►▼
Show Figures
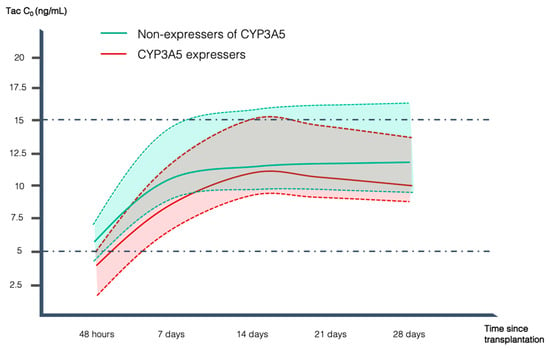
Figure 1

Journal Menu
► ▼ Journal Menu-
- Pharmaceuticals Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
22 April 2024
Pharmaceuticals | Top 5 Selected Papers in 2022–2023 in the Section “Medicinal Chemistry”
Pharmaceuticals | Top 5 Selected Papers in 2022–2023 in the Section “Medicinal Chemistry”

19 April 2024
Pharmaceuticals | Top 5 Selected Papers in 2022–2023 in the Section “Pharmaceutical Technology”
Pharmaceuticals | Top 5 Selected Papers in 2022–2023 in the Section “Pharmaceutical Technology”

Topics
Topic in
Antioxidants, IJPB, Molecules, Pharmaceuticals, Plants
Plants Volatile Compounds
Topic Editors: Dario Kremer, Igor Jerković, Valerija DunkićDeadline: 30 April 2024
Topic in
BioMed, Biomedicines, Osteology, Pharmaceuticals, Pharmaceutics
Recent Advancement in Biotechnology and Drug Development Using Cutting-Edge Platforms
Topic Editors: Hani Al-Salami, Armin MooranianDeadline: 31 May 2024
Topic in
Antibiotics, JPM, Pharmaceuticals, Pharmaceutics
Pharmacokinetic and Pharmacodynamic Modelling in Drug Discovery and Development
Topic Editors: Inaki F. Troconiz, Victor Mangas Sanjuán, Maria Garcia-Cremades MiraDeadline: 31 August 2024
Topic in
Cancers, Diseases, Nanomaterials, Pharmaceuticals, Materials
Nanomaterials and Diseases
Topic Editors: Diana Díaz-García, Santiago Gómez-RuizDeadline: 13 October 2024

Conferences
Special Issues
Special Issue in
Pharmaceuticals
Current Frames on Cardiotoxicology of New and Old Anticancer Drugs
Guest Editor: Vera Marisa CostaDeadline: 8 May 2024
Special Issue in
Pharmaceuticals
Therapeutic Target Developments in Pancreatic Cancer
Guest Editors: Guishan Xiao, Bin LuDeadline: 12 May 2024
Special Issue in
Pharmaceuticals
Recent Advances in TGF-β Inhibitors for the Therapeutic Management of Cancer and Fibrosis
Guest Editor: David DaniepourDeadline: 31 May 2024
Special Issue in
Pharmaceuticals
Structure and Ligand Based Drug Design
Guest Editor: Dongsheng CaoDeadline: 10 June 2024
Topical Collections
Topical Collection in
Pharmaceuticals
Old Pharmaceuticals with New Applications
Collection Editor: Massimiliano Tognolini
Topical Collection in
Pharmaceuticals
Drug Discovery and Development for Tropical Diseases (TDs)
Collection Editor: Christophe Dardonville
Topical Collection in
Pharmaceuticals
The Story of Successful Drugs and Recent FDA-Approved Molecules
Collection Editors: Maria Emília De Sousa, Guangshun Wang, Jean Jacques Vanden Eynde, Klaus Kopka, Annie Mayence, Joachim JoseConference Reports
Pharmaceuticals 2023, 16(3), 432; https://doi.org/10.3390/ph16030432
Pharmaceuticals 2022, 15(4), 388; https://doi.org/10.3390/ph15040388